You have no items in your shopping cart.
All Products
- Featured
 Human SPI1 protein [orb419162]Featured
Human SPI1 protein [orb419162]Featured
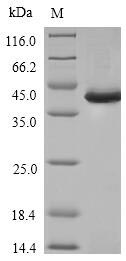
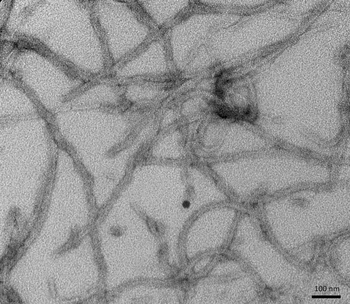
Greater than 85% as determined by SDS-PAGE.
35.1 kDa
E.coli
20 μg, 100 μg, 1 mg SDS-PAGE
Unconjugated
Human IgG Fab fragment was prepared from normal serum by a multi-step process which includes delipidation, salt fractionation and ion exchange chromatography followed by papain digestion and extensive dialysis against the buffer stated above. Human IgG Fab fragment assayed by immunoelectrophoresis resulted in a single precipitin arc against anti-Human Serum, anti- Human IgG and anti- Human IgG F(ab’)2. No reaction was observed against anti- Human IgG F(c) or anti-Papain.
Human
2 mg- Featured
 Featured
Featured
Unconjugated
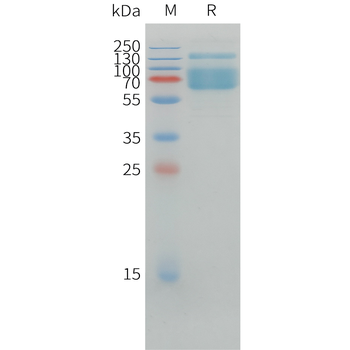
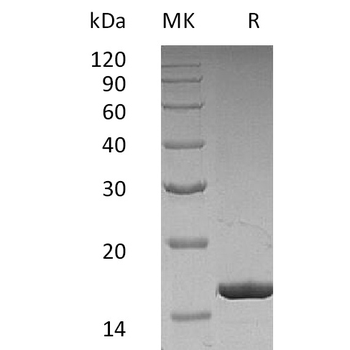
Greater than 95% as determined by reducing SDS-PAGE.
16.6 KDa
Mammalian
10 μg, 50 μg - Featured
 Featured
Featured
In vitro, In vivo, WB
>98%
4.5 kDa
Synthetic
100 μg - Featured
 Featured
Featured
Unconjugated
The purity of the protein is greater than 95% as determined by SDS-PAGE and Coomassie blue staining.
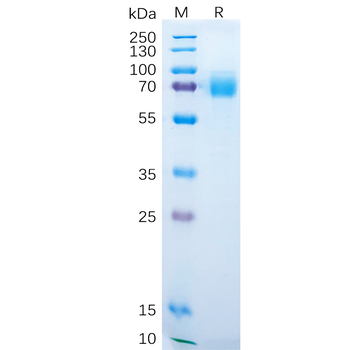
The protein has a predicted molecular mass of 36.8 kDa after removal of the signal peptide. The apparent molecular mass of ACVRL1-hFc is approximately 35-70 kDa due to glycosylation.
E.coli
100 μg, 10 μg, 50 μg - Featured
 Featured
Featured
Unconjugated
The purity of the protein is greater than 95% as determined by SDS-PAGE and Coomassie blue staining.
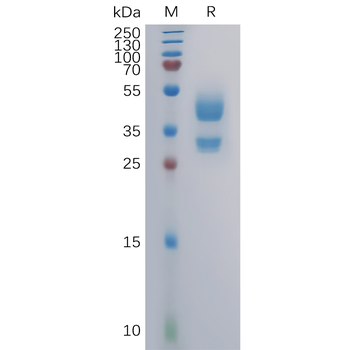
The protein has a predicted molecular mass of 10.4 and 37.9 kDa after removal of the signal peptide. The apparent molecular mass of CD3D-His and CD3E-hFc is approximately 35-55 kDa due to glycosylation.
Mammalian
10 μg, 50 μg, 100 μg - Featured
 Featured
Featured
Unconjugated
The purity of the protein is greater than 95% as determined by SDS-PAGE and Coomassie blue staining.
The protein has a predicted molecular mass of 50.5 kDa after removal of the signal peptide. The apparent molecular mass of hFc-FZD7 is approximately 55-70 kDa due to glycosylation.
Mammalian
10 μg, 50 μg, 100 μg - Featured
 Featured
Featured
Unconjugated
The purity of the protein is greater than 95% as determined by SDS-PAGE and Coomassie blue staining.
The protein has a predicted molecular mass of 39.4 kDa after removal of the signal peptide. The apparent molecular mass of hFc-S100A9 is approximately 35-55 kDa due to glycosylation.
E.coli
10 μg, 50 μg, 100 μg - Featured
 Featured
Featured
In vitro, In vivo, SDS-PAGE, WB
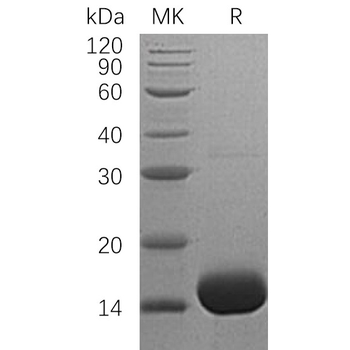
>95%
~15.1 kDa
Recombinant
100 μg