You have no items in your shopping cart.
Cart summary

Factor V antibody
Catalog Number: orb21223
| Catalog Number | orb21223 |
|---|---|
| Category | Antibodies |
| Description | Precipitating polyclonal rabbit antiserum to human coagulation factor V. Plasma factor V is a relatively labile glycoprotein (MW 350,000) which is essential for normal clotting and haemostasis. It is synthesized in hepatocytes and reticuloendothelial cells in the liver. Factor V is present in platelet alpha-granules but not on the surface of the intact platelet. It is released following platelet aggregation and its coagulant activity is distinguishable from plasma factor V. Thrombin activates Factor V to FVa by proteolysis resulting in the release of several polypeptides with molecular weight of 70,000 to 150,000. After clotting he protein is no longer detectable in the serum. FVa binds to receptor sites in the platelet membrane which protects Fva from the action of inhibitors (e.g. protein C). The concentration of factor V in adult plasma is 1-3 mg/ml. Newborn infants have similar levels. Factor V deficiency is associated with severe haemorrhagic disorder. Congenital deficiency with an autosomal bleeding is relatively rare. It exists in two molecular forms: coagulant activity may be reduced together with factor V antigen levels (impaired synthesis), or low coagulant activity is associated with the presence of a variable level of plasma factor V (abnormal molecules). Both conditions lead to frequent minor bleedings of skin and mucosal tissues. Acquired deficiency with reduced factor V antigen levels can be a reliable parameter of liver damage in severe liver diseases. Circulating antibodies to factor V acting as neutralizing inhibitors of plasma an platelet factor V have been described. Highly purified Factor V is isolated from pooled human plasma and used for immunization. Freund's complete adjuvant is used in the first step of the immunization procedure. |
| Species/Host | Rabbit |
| Clonality | Polyclonal |
| Form/Appearance | Delipidated, heat inactivated, lyophilized, stable whole serum, dialyzed against glycine buffer. Sodium azide 1 mg/ml Total protein and IgG concentrations in the antiserum are comparable to those of pooled normal rabbit serum. No foreign proteins added. |
| Conjugation | Unconjugated |
| Hazard Information | This product is intended FOR RESEARCH USE ONLY, and FOR TESTS IN VITRO, not for use in diagnostic or therapeutic procedures involving humans or animals. This product contains sodium azide. To prevent formation of toxic vapors, do not mix with strong acidic solutions. To prevent formation of potentially explosive metallic azides in metal plumbing, always wash into drain with copious quantities of water. This datasheet is as accurate as reasonably achievable, but Nordic-MUbio accepts no liability for any inaccuracies or omissions in this information. |
| UniProt ID | P12259 |
| Source | Plasma factor V is a relatively labile glycoprotein (MW 350,000) which is essential for normal clotting and haemostasis. It is synthesized in hepatocytes and reticuloendothelial cells in the liver. Factor V is present in platelet alpha-granules but not on the surface of the intact platelet. It is released following platelet aggregation and its coagulant activity is distinguishable from plasma factor V. Thrombin activates Factor V to FVa by proteolysis resulting in the release of several polypeptides with molecular weight of 70,000 to 150,000. After clotting he protein is no longer detectable in the serum. FVa binds to receptor sites in the platelet membrane which protects Fva from the action of inhibitors (e.g. protein C). The concentration of factor V in adult plasma is 1-3 μg/ml. Newborn infants have similar levels. Factor V deficiency is associated with severe haemorrhagic disorder. Congenital deficiency with an autosomal bleeding is relatively rare. It exists in two molecular forms: coagulant activity may be reduced together with factor V antigen levels (impaired synthesis), or low coagulant activity is associated with the presence of a variable level of plasma factor V (abnormal molecules). Both conditions lead to frequent minor bleedings of skin and mucosal tissues. Acquired deficiency with reduced factor V antigen levels can be a reliable parameter of liver damage in severe liver diseases. Circulating antibodies to factor V acting as neutralizing inhibitors of plasma an platelet factor V have been described. Highly purified Factor V is isolated from pooled human plasma and used for immunization. Freund’s complete adjuvant is used in the first step of the immunization procedure. |
| Storage | Storage: The lyophilized antiserum is shipped at ambient temperature and may be stored at +4°C; prolonged storage at or below -20°C. Dilutions may be prepared by adding phosphate buffered saline (PBS, pH 7.2). Repeated thawing and freezing should be avoided. If a slight precipitation occurs upon storage, this should be removed by centrifugation. It will not affect the performance of the antiserum. Diluted antiserum should be stored at +4°C, not refrozen, and preferably used the same day |
| Buffer/Preservatives | Delipidated, heat inactivated, lyophilized, stable whole serum, dialyzed against glycine buffer. Sodium azide 1 mg/ml Total protein and IgG concentrations in the antiserum are comparable to those of pooled normal rabbit serum. No foreign proteins added. |
| Note | For research use only |
| Expiration Date | 12 months from date of receipt. |
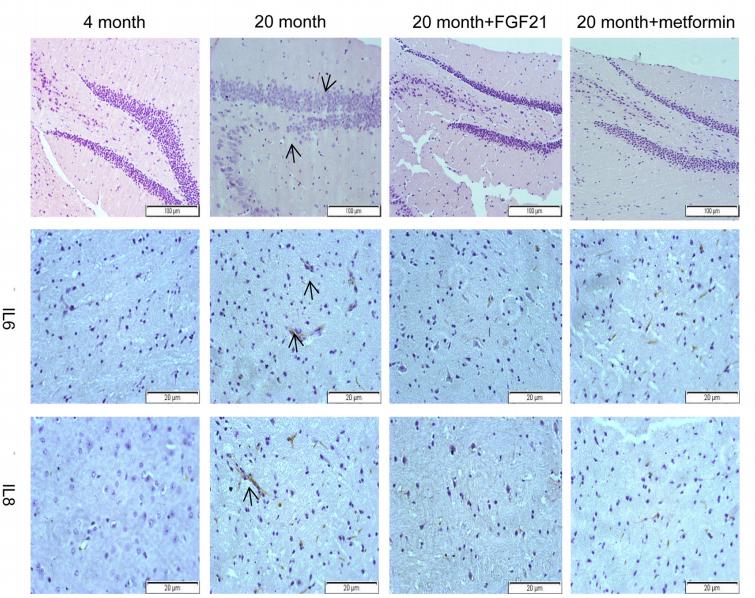
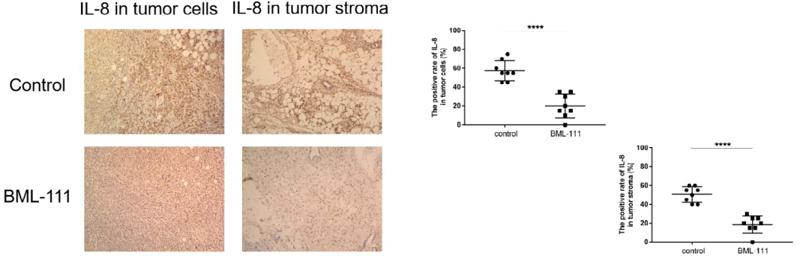
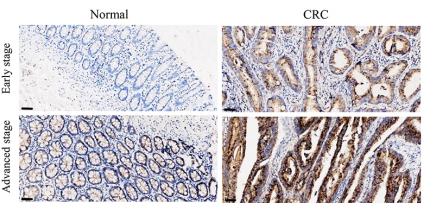
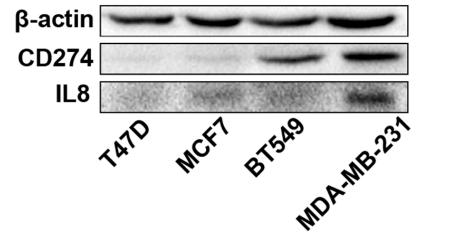
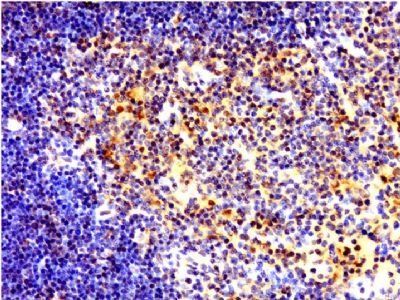
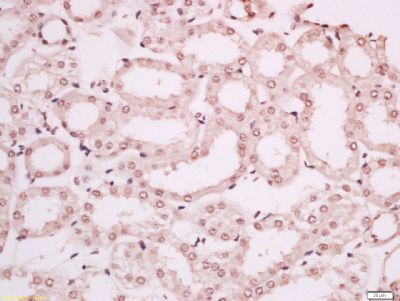
IL8 antibody [orb229133]
IHC-P, WB
Human, Mouse, Rat
Rabbit
Polyclonal
Unconjugated
100 μl, 50 μl, 200 μlc-Maf/MAF Antibody [orb614126]
FC, IHC, WB
Human, Mouse, Rat
Rabbit
Polyclonal
Unconjugated
10 μg, 100 μg
Submit a review
Filter by Rating
- 5 stars
- 4 stars
- 3 stars
- 2 stars
- 1 stars