You have no items in your shopping cart.
Cart summary

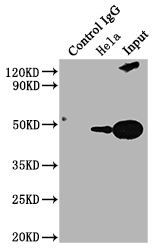
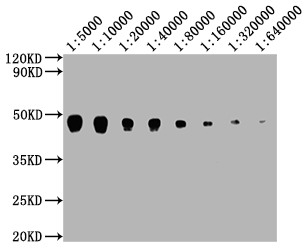
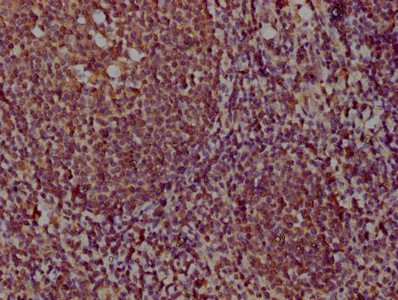
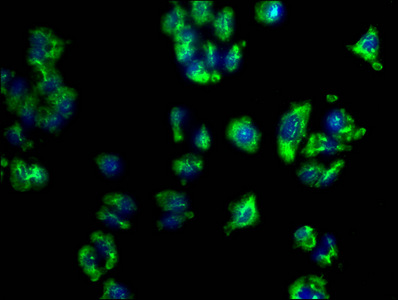
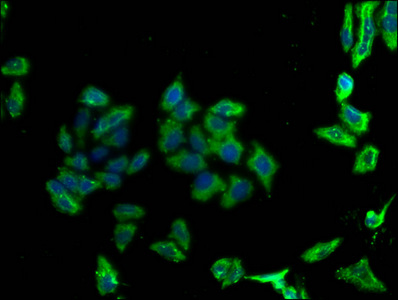
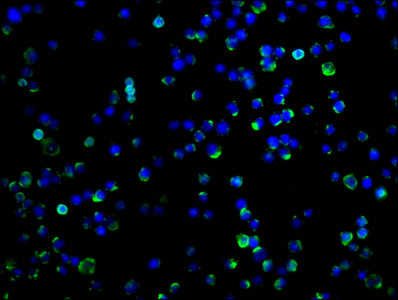
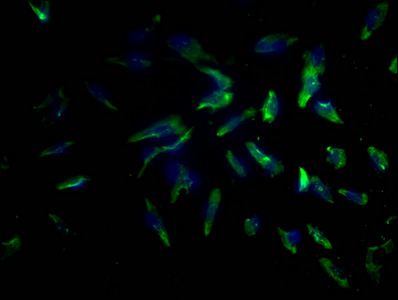
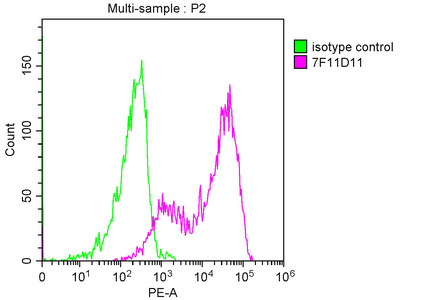
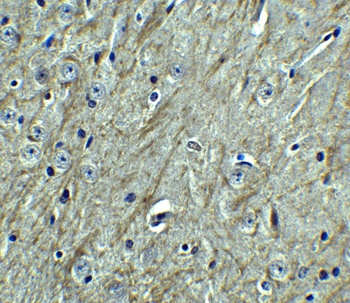
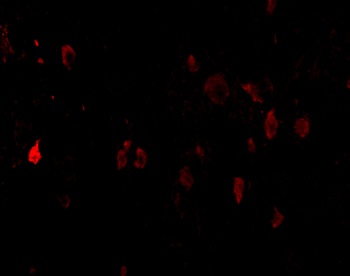
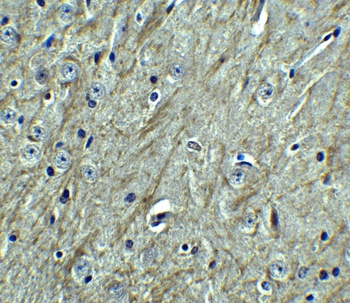
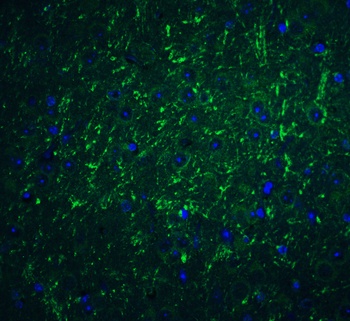
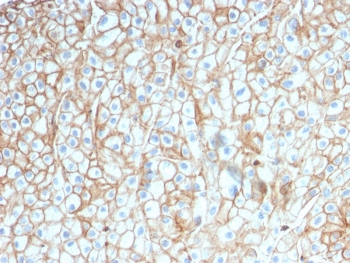
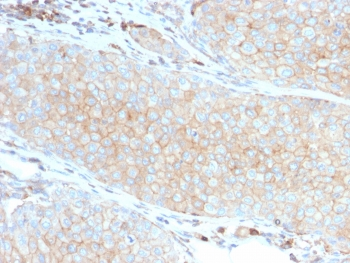
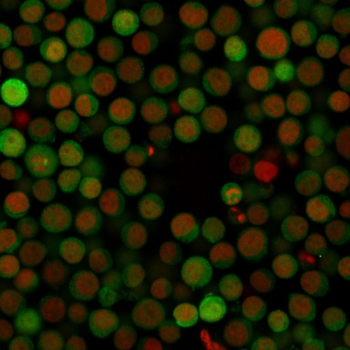
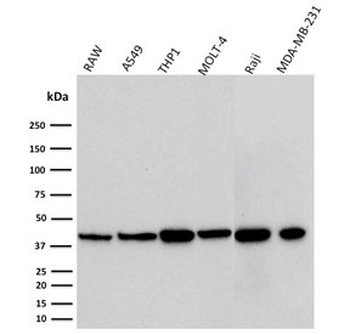
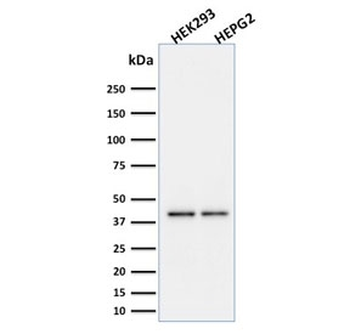
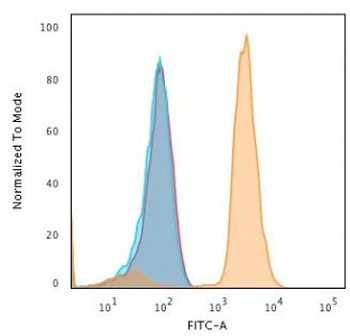
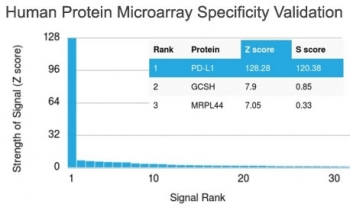
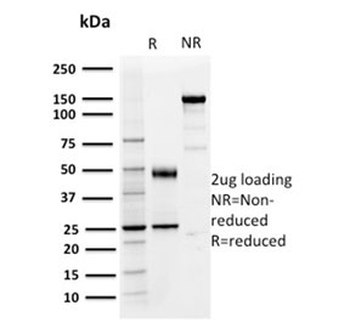
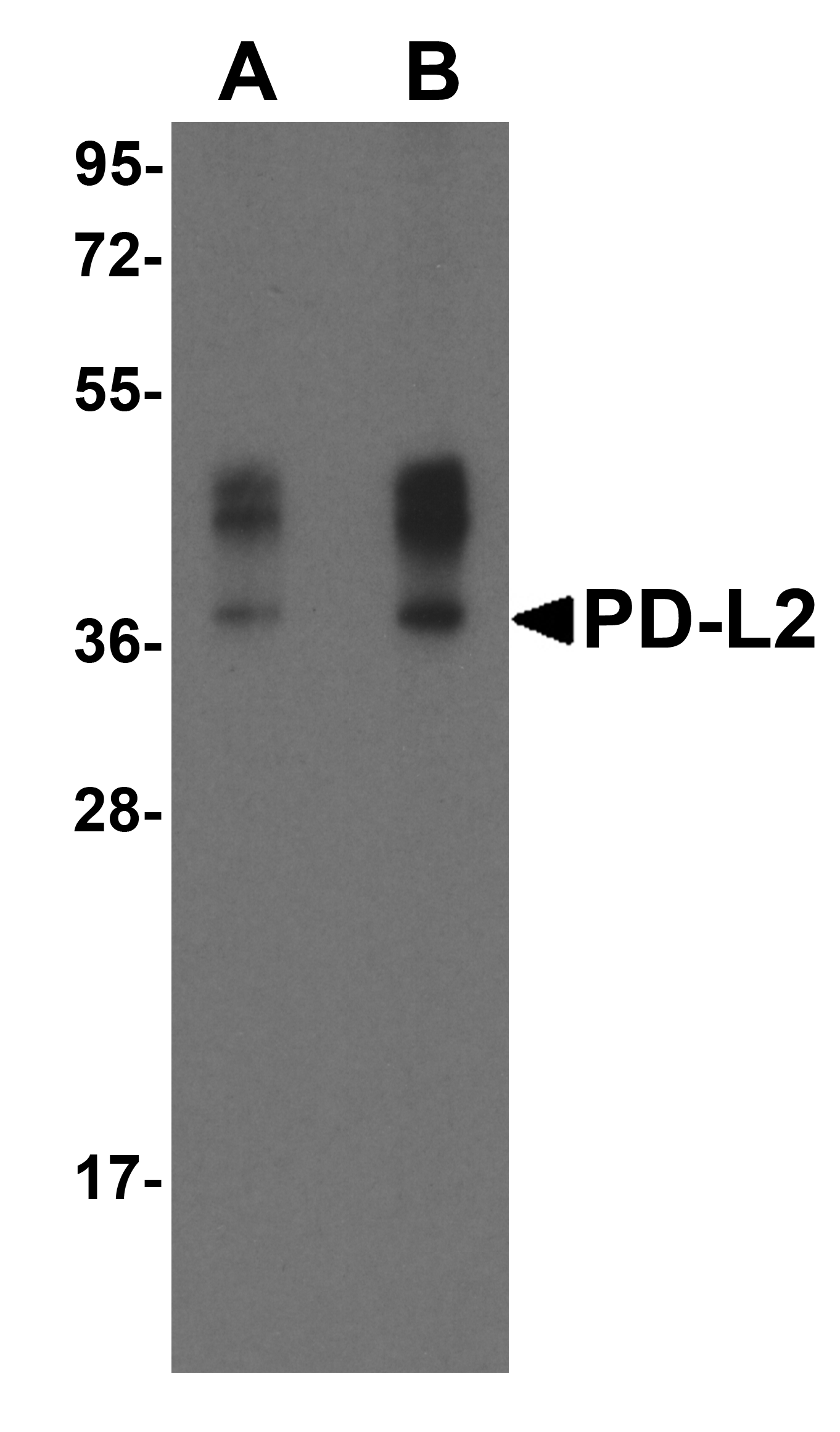
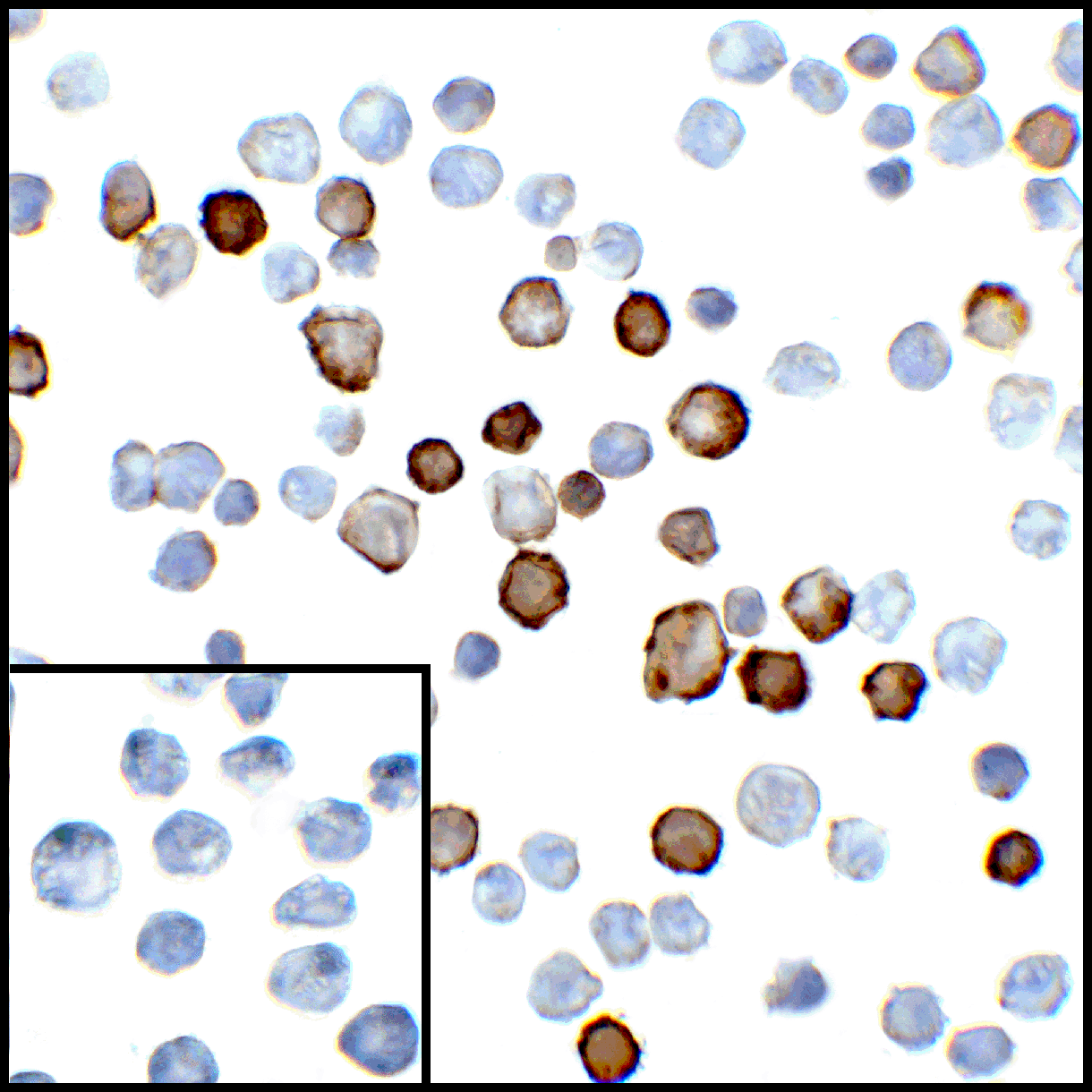
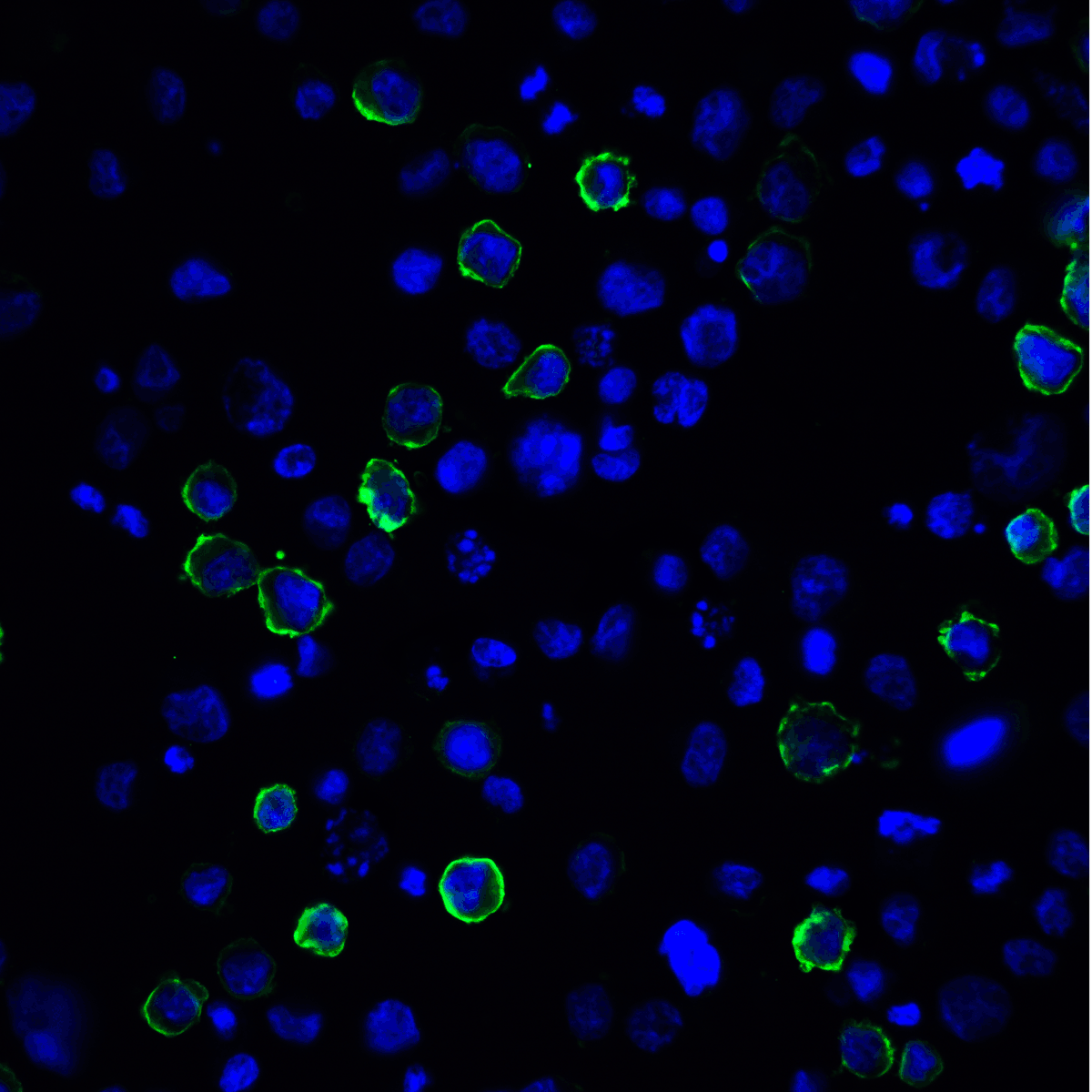
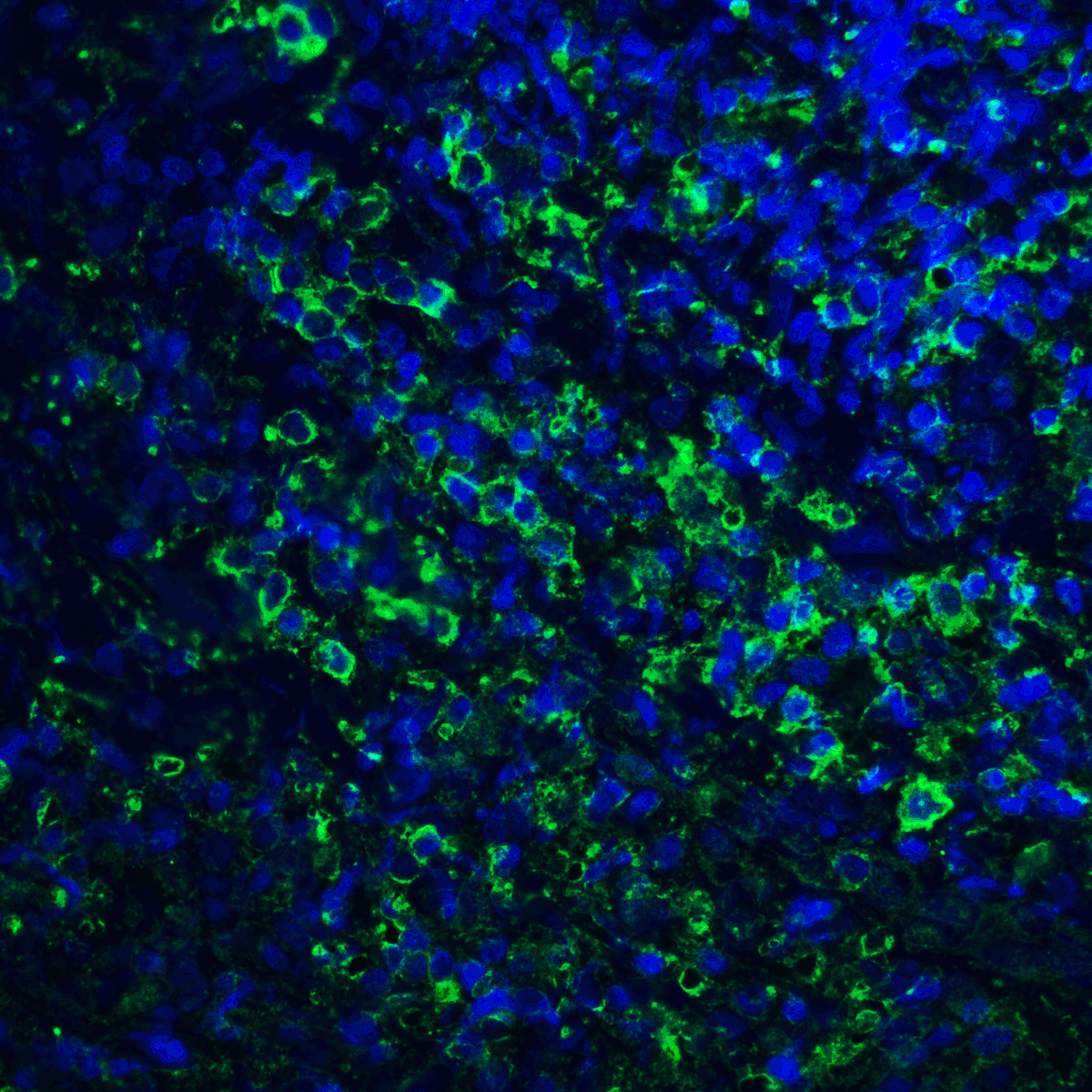
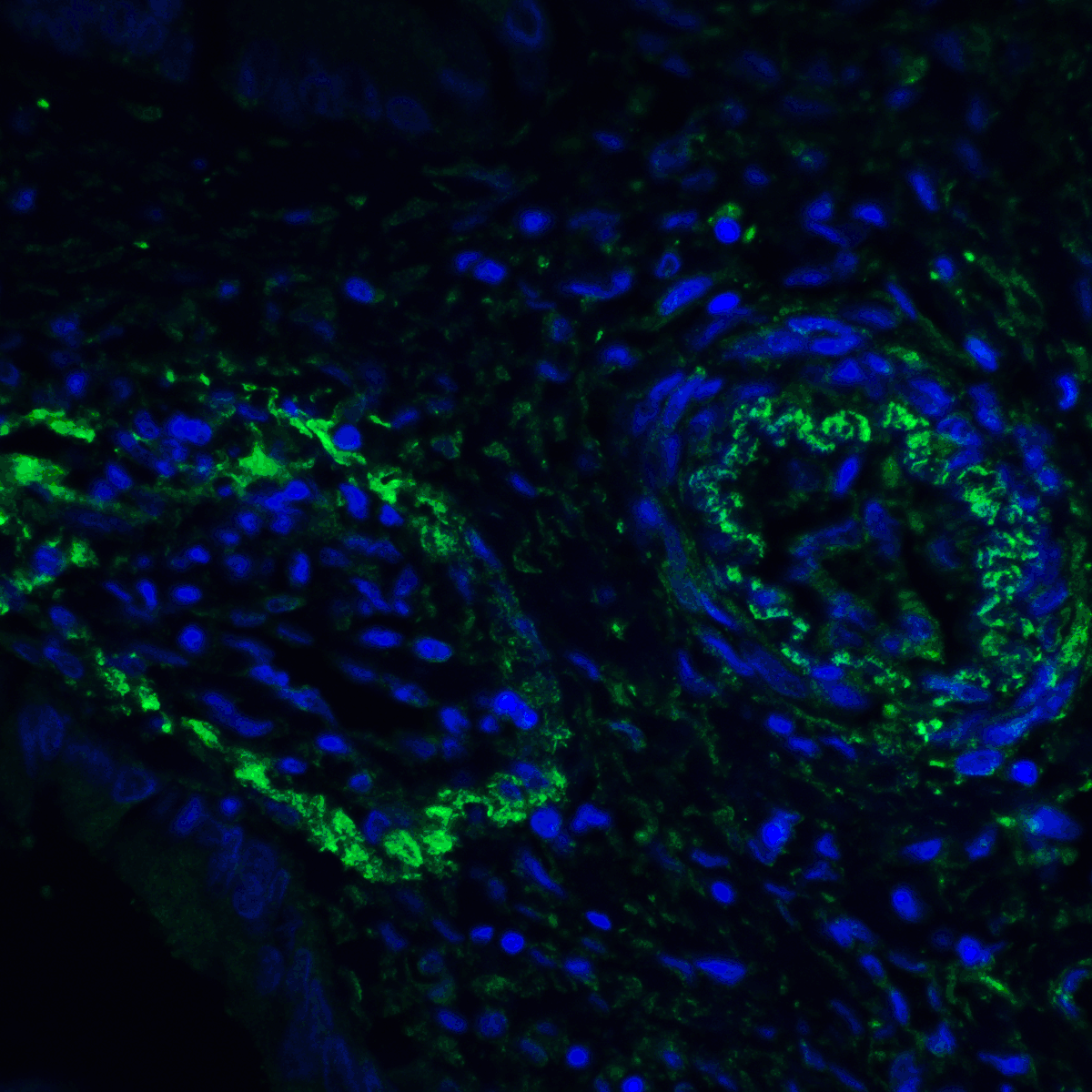
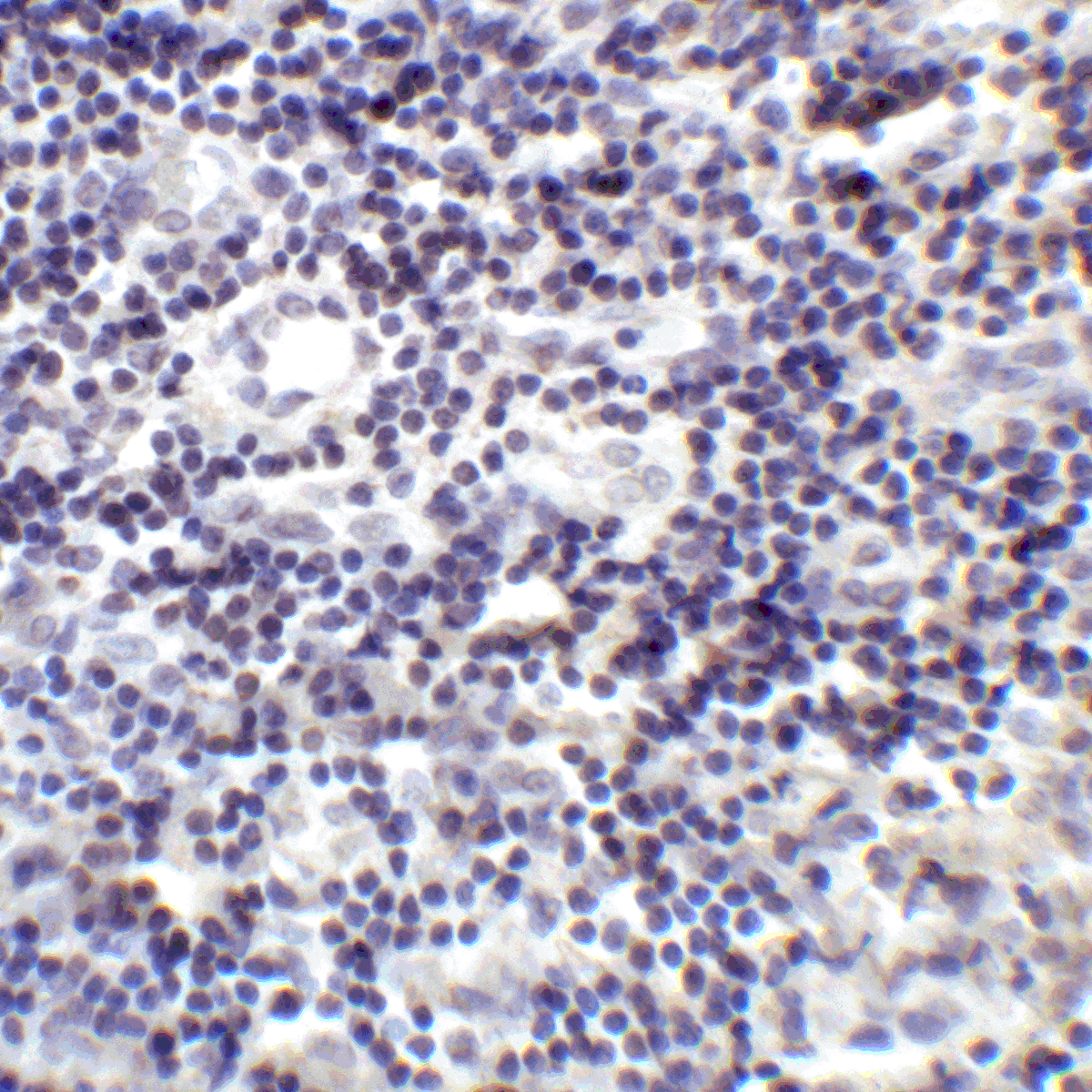
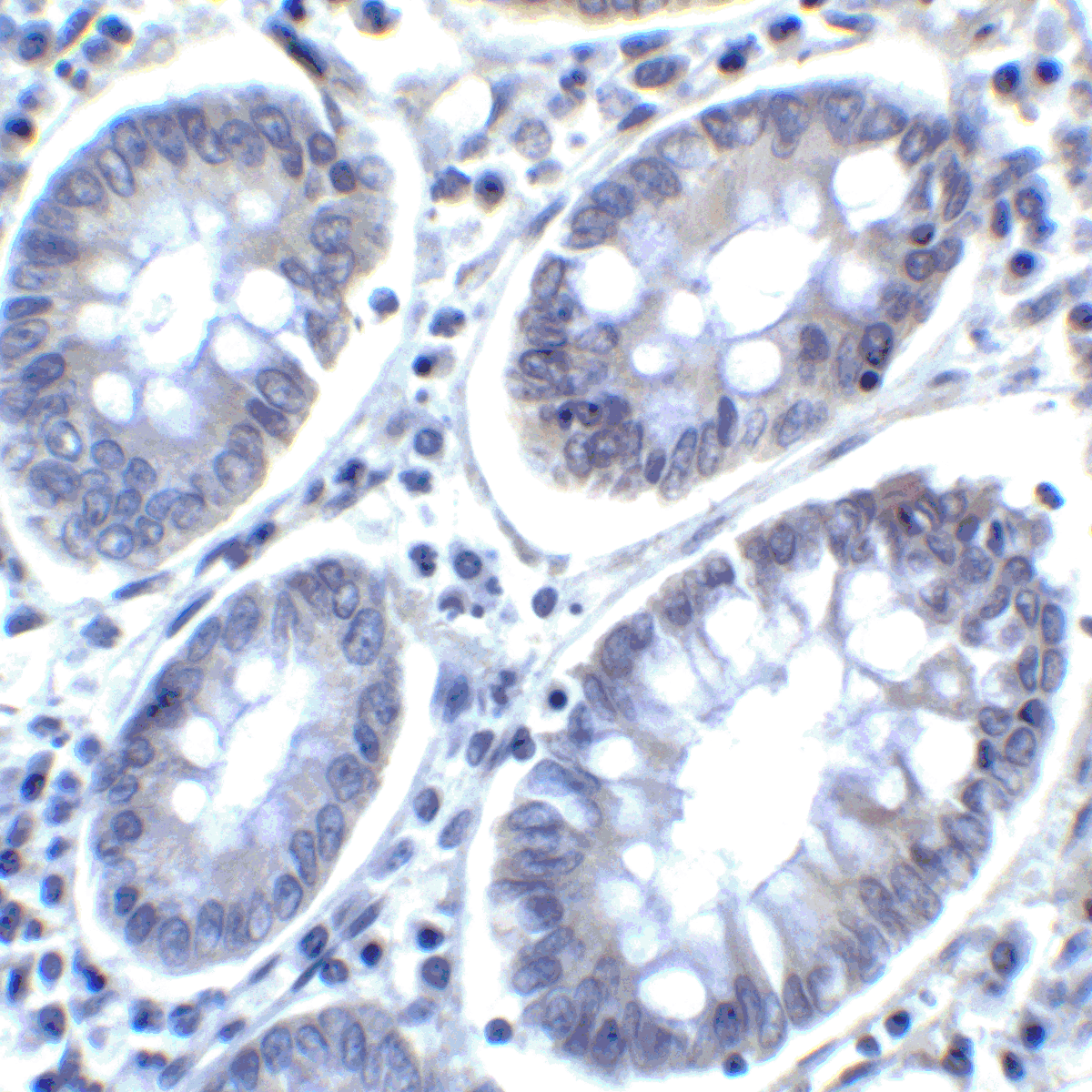
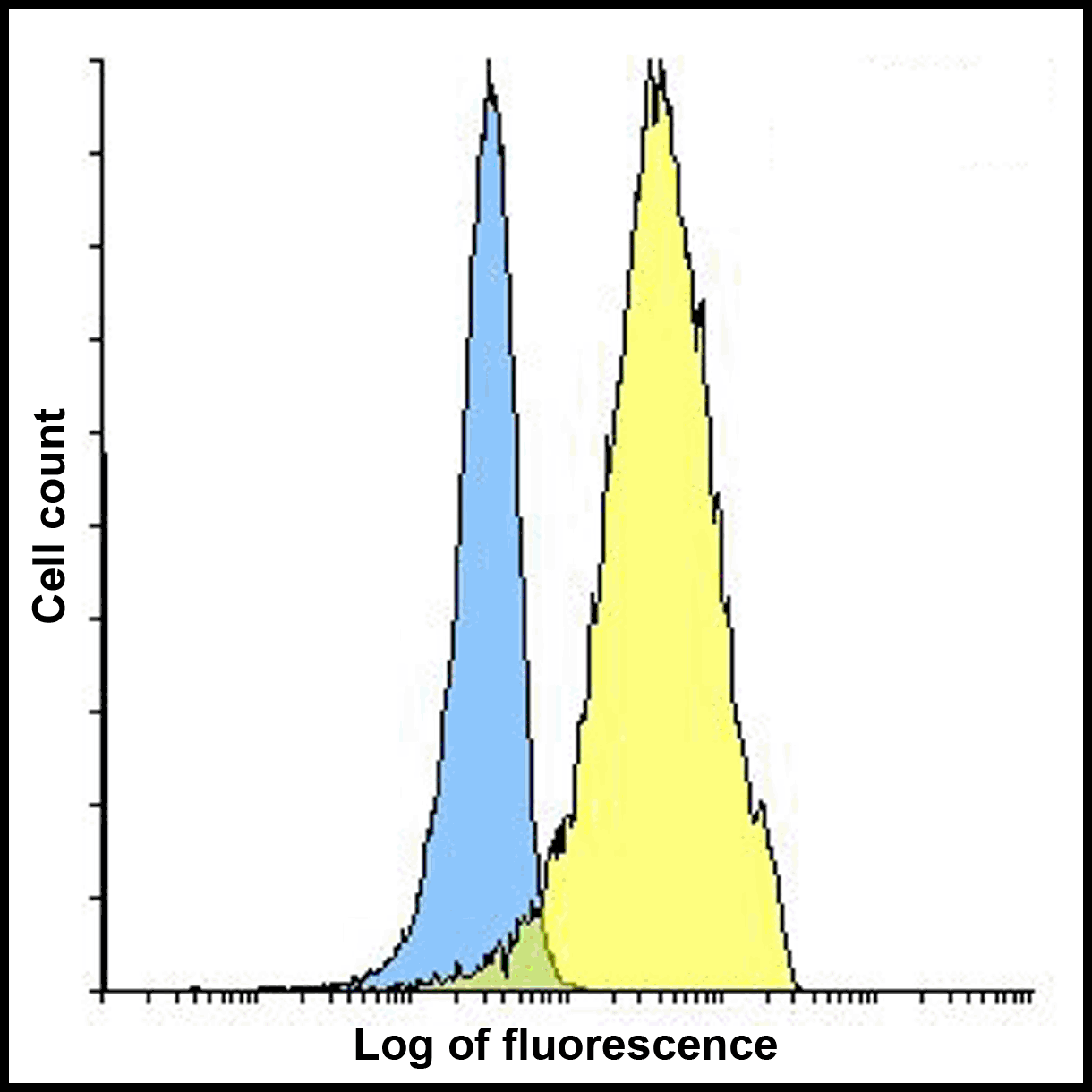
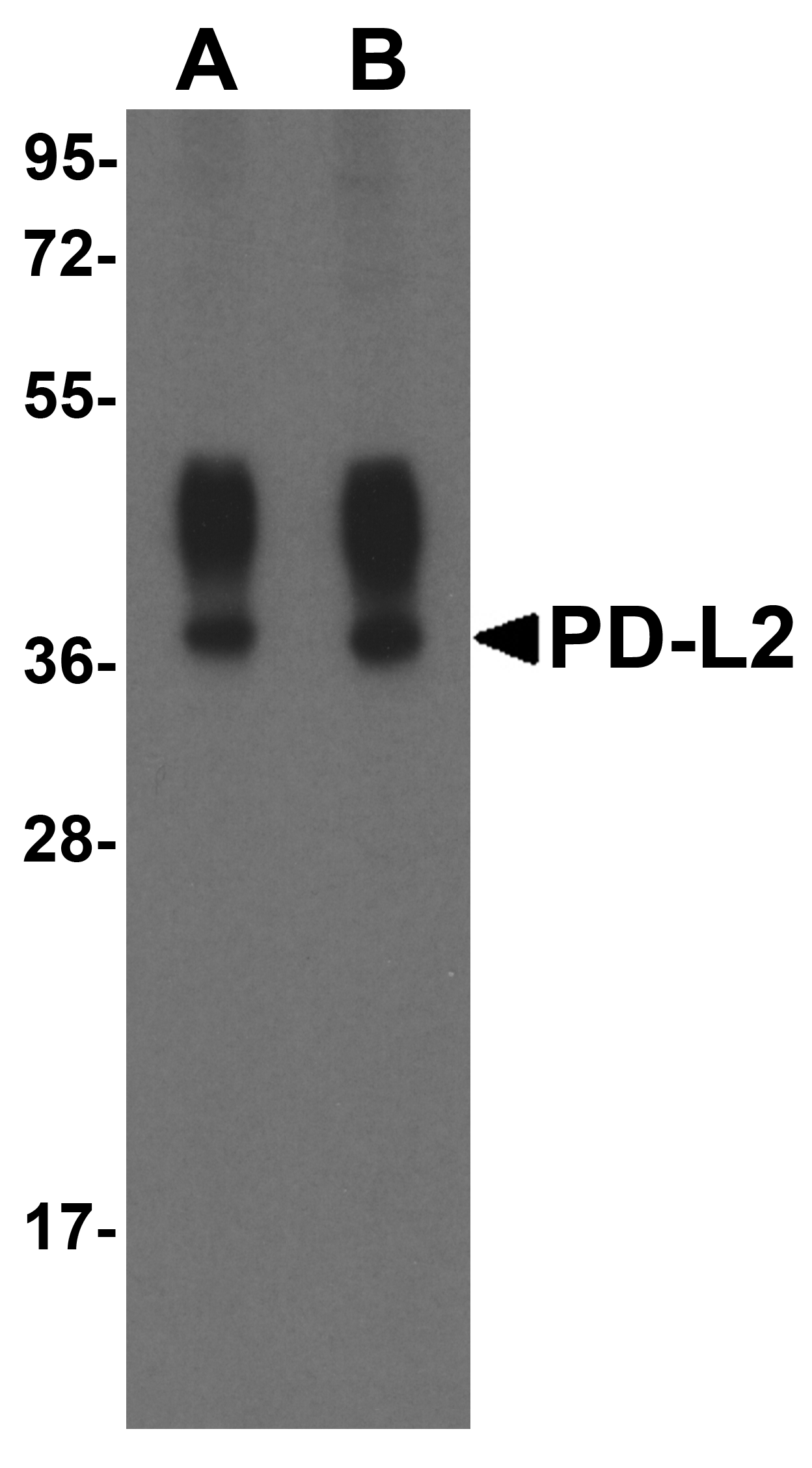
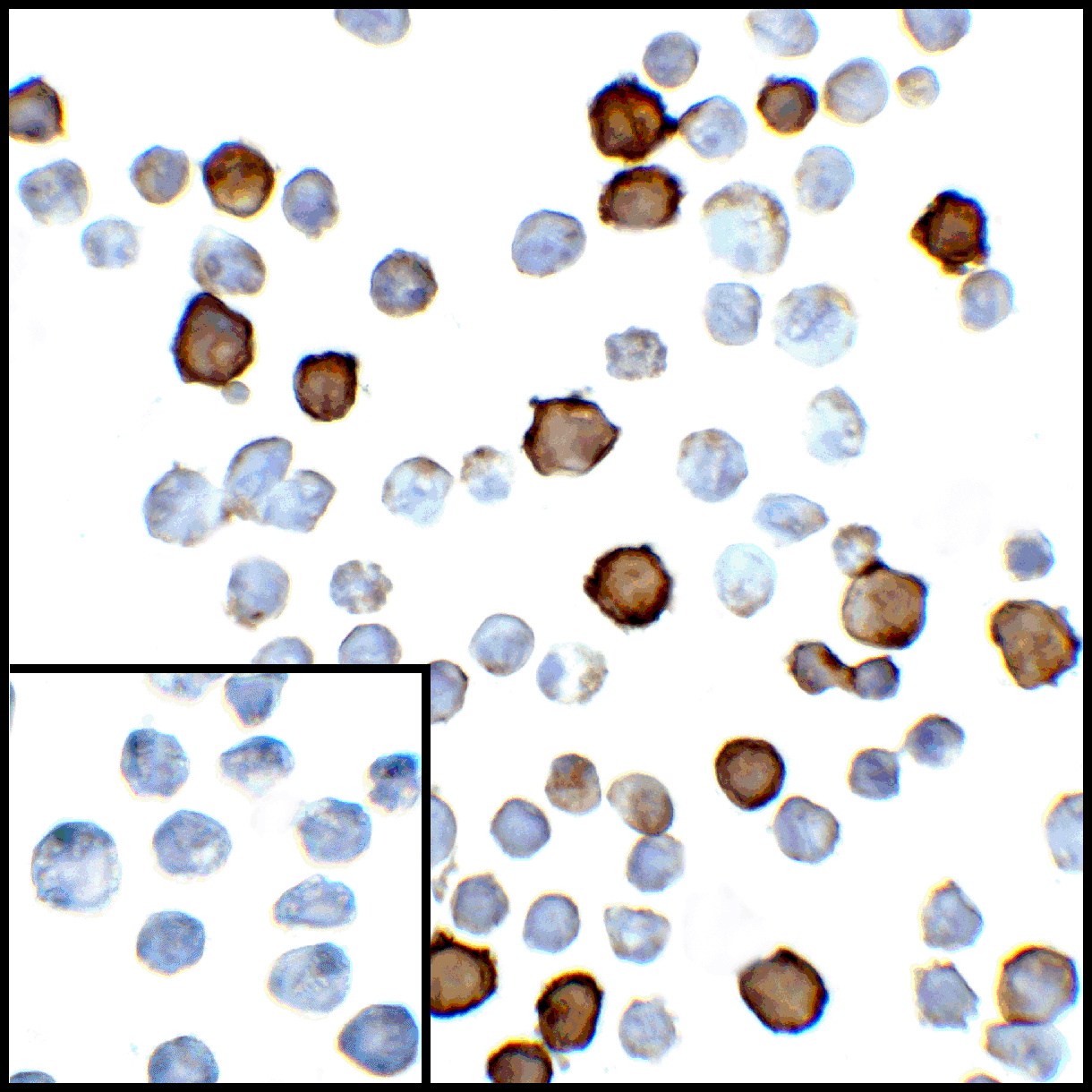
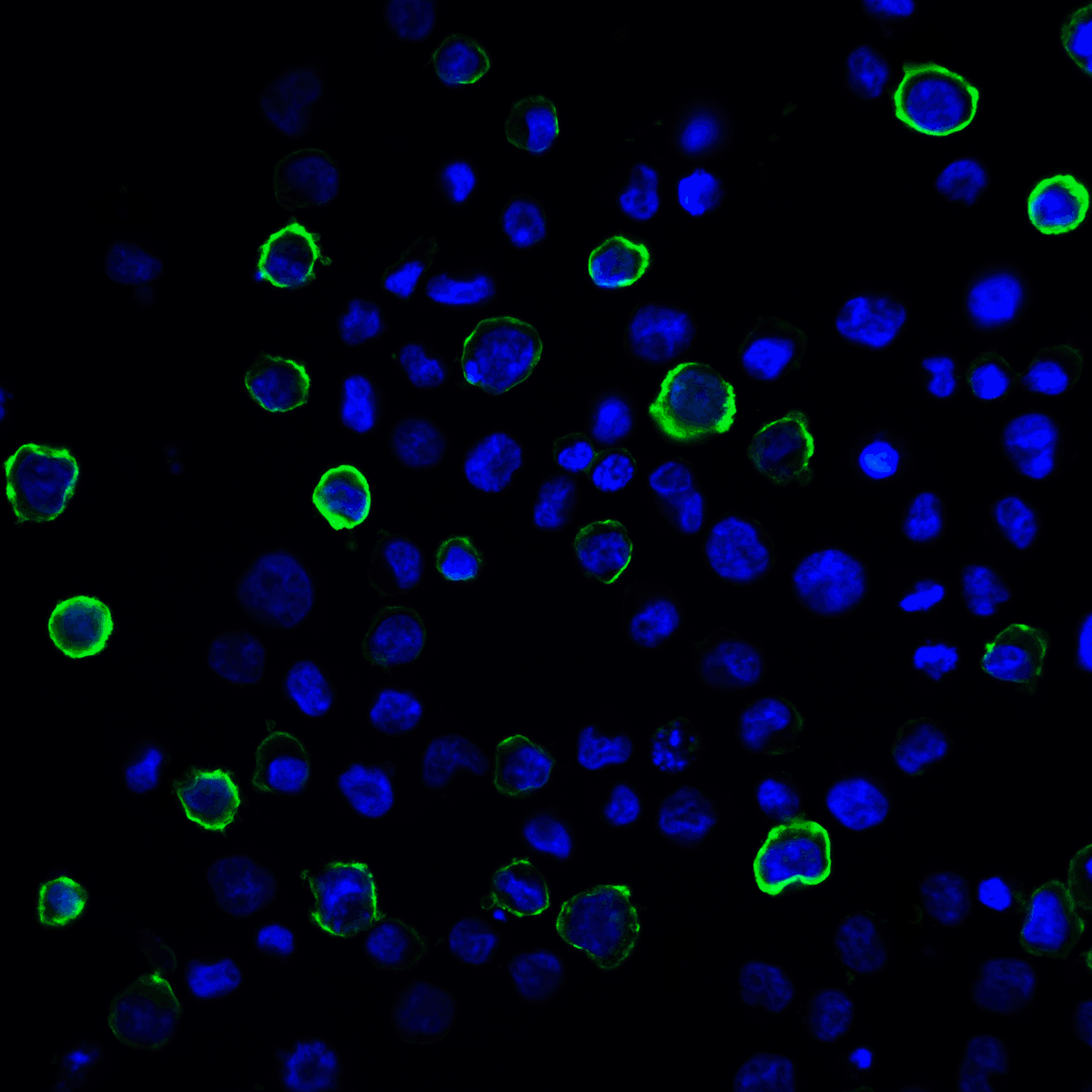
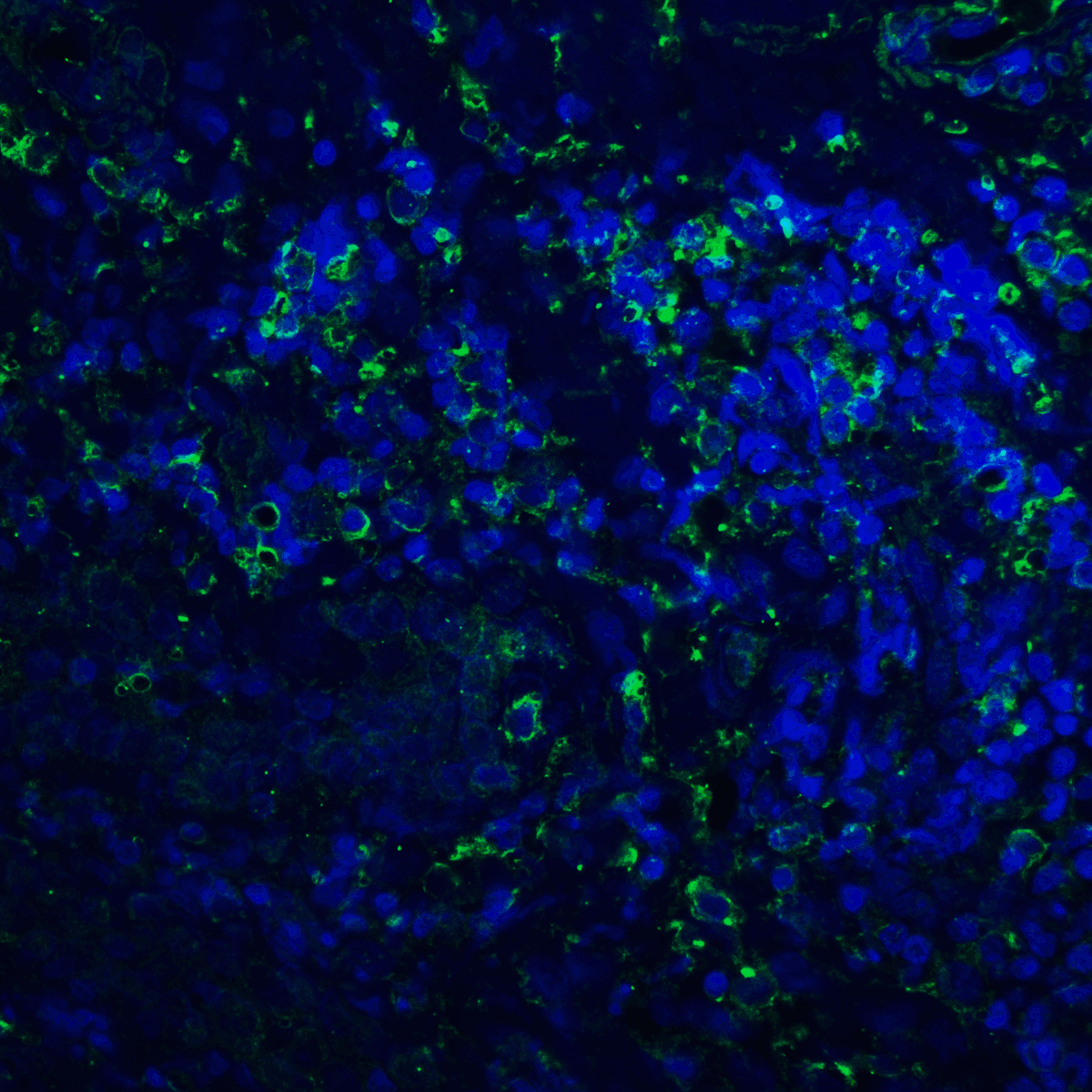
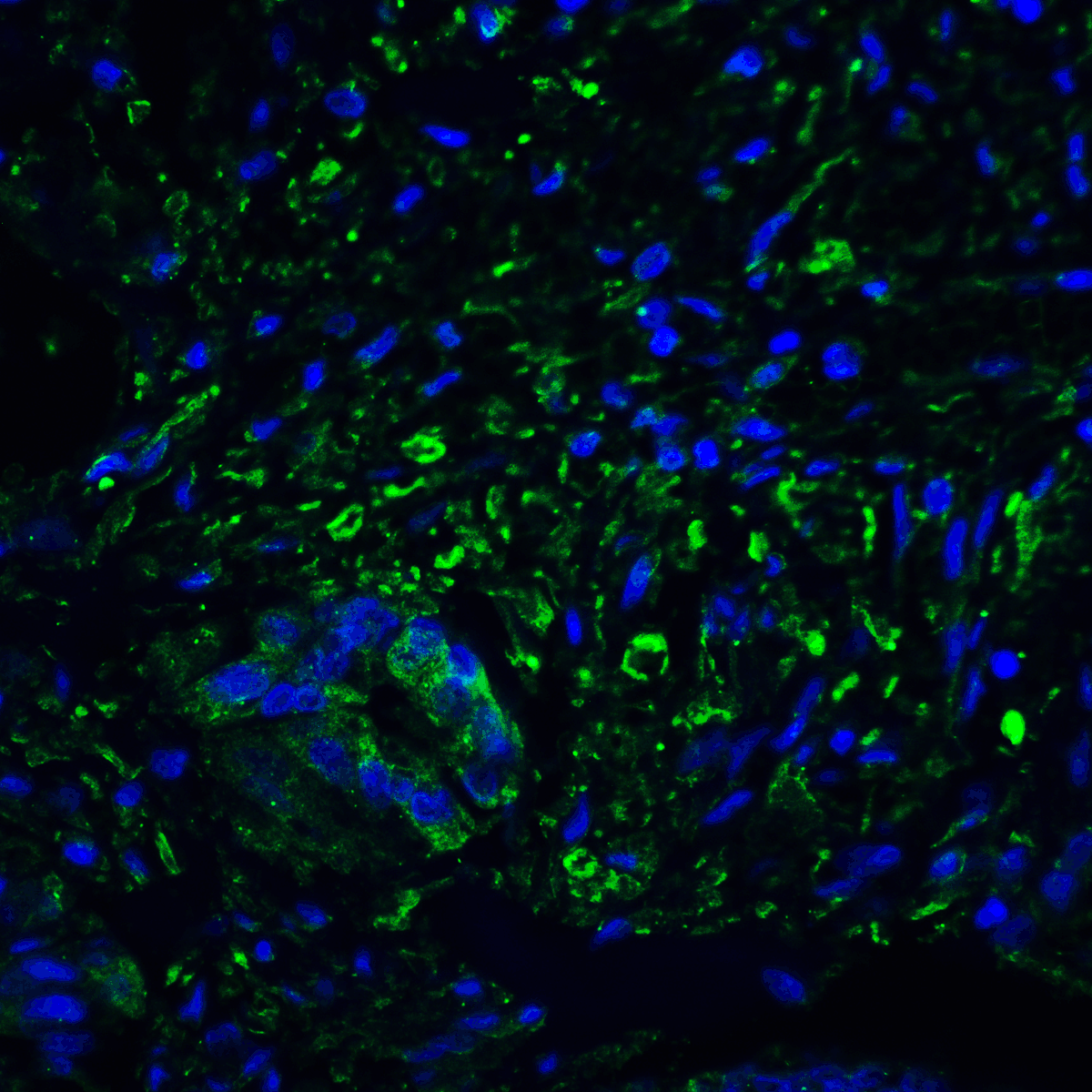
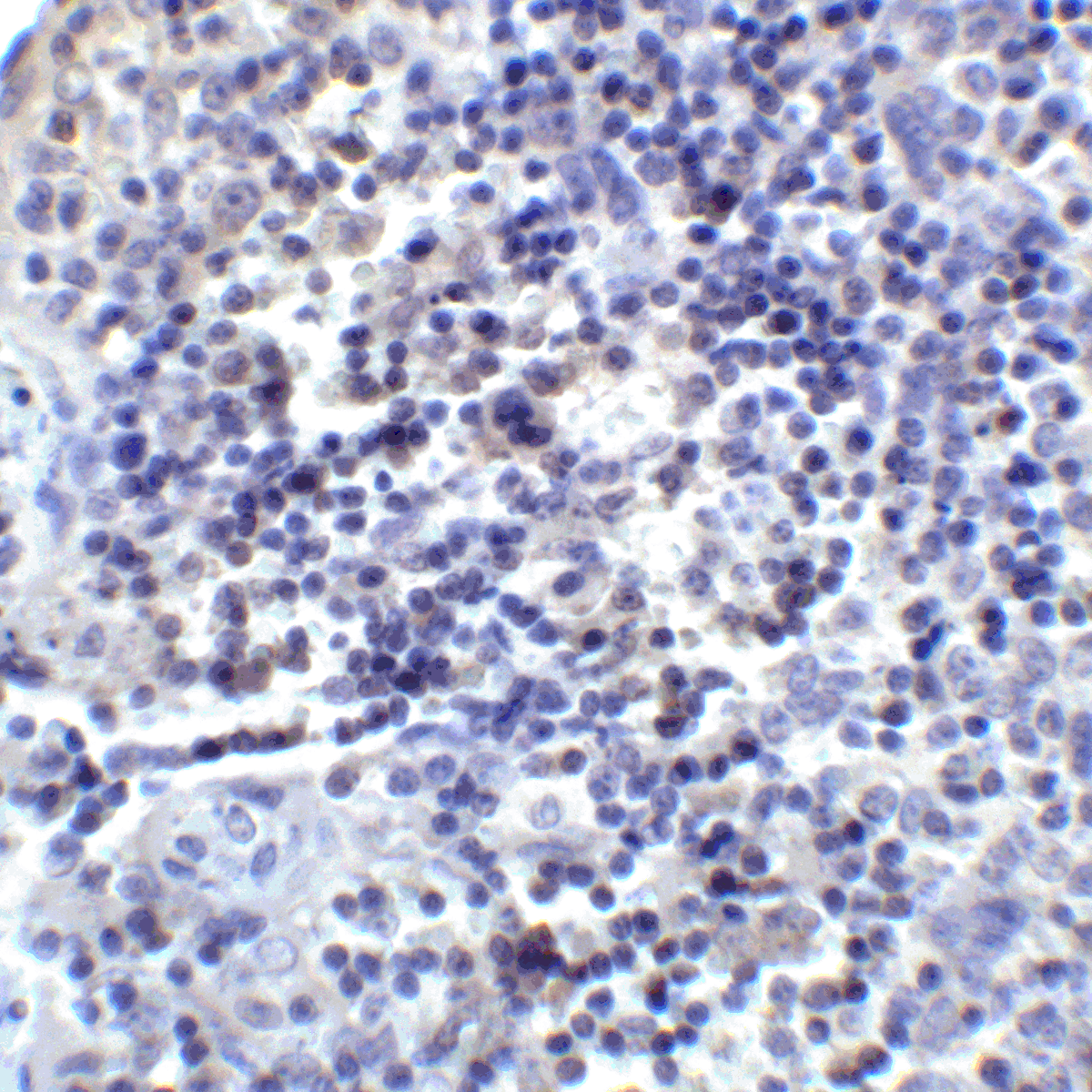
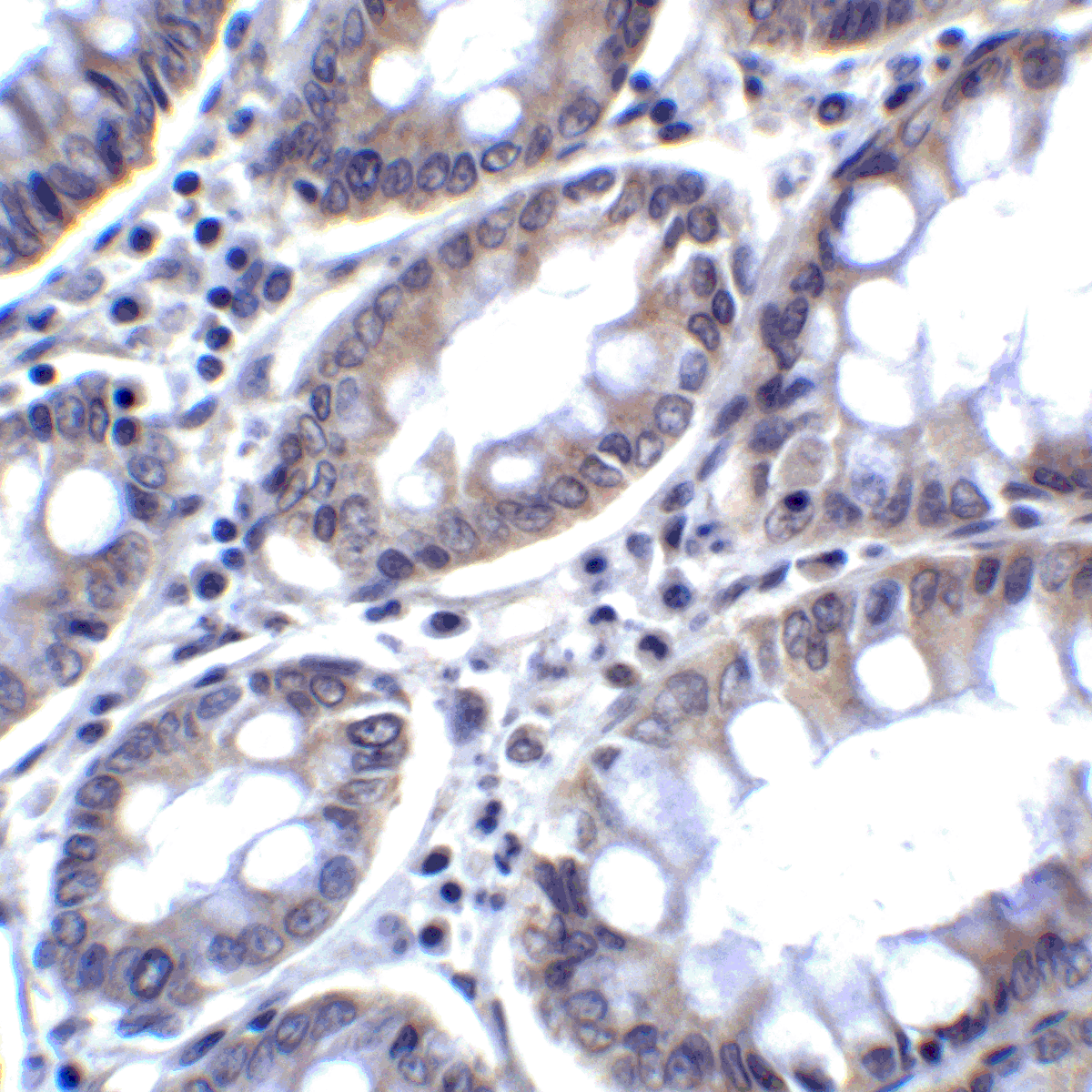
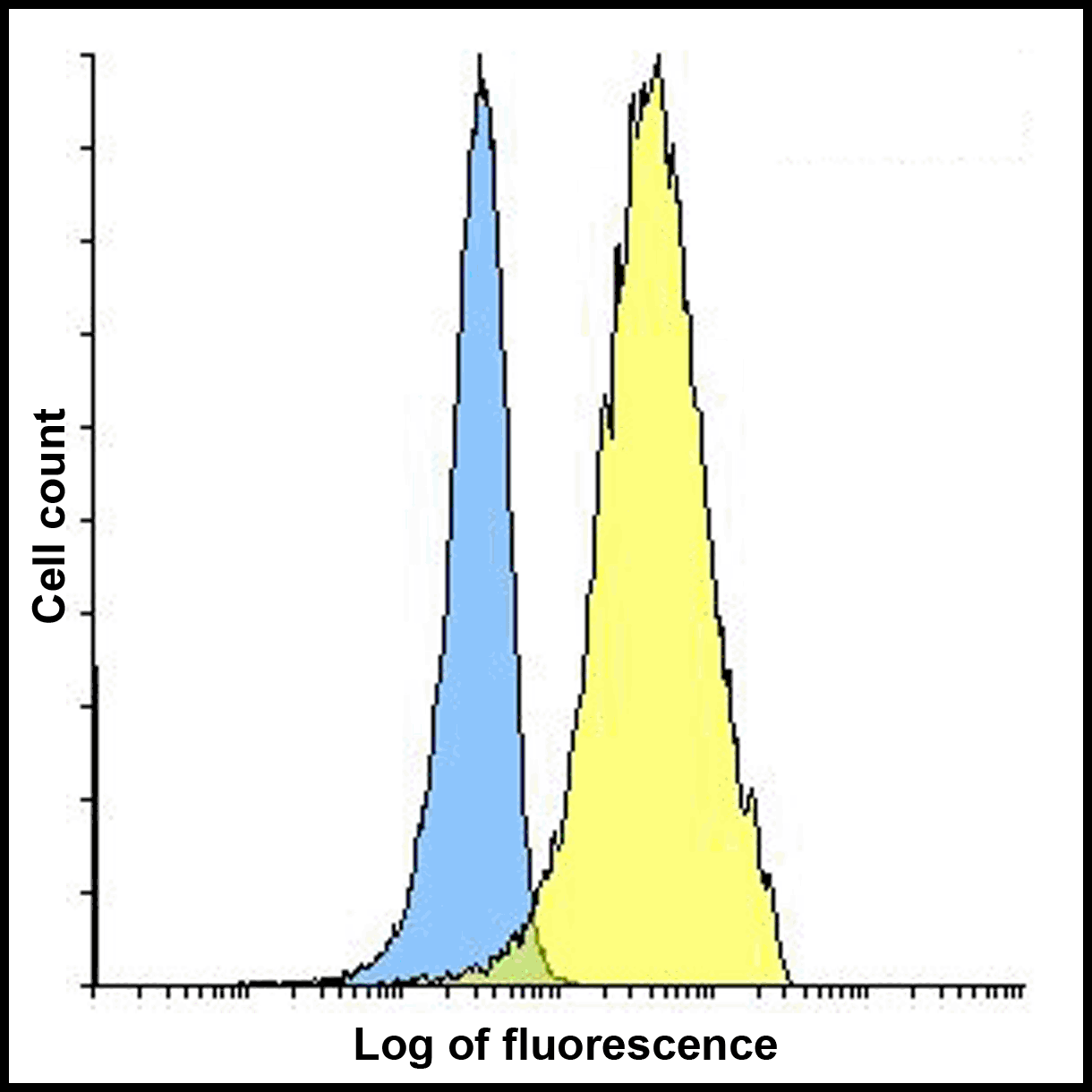
PD-1 Antibody
Catalog Number: orb1821655
| Catalog Number | orb1821655 |
|---|---|
| Category | Antibodies |
| Description | PD-1 Antibody |
| Species/Host | Human |
| Clonality | Monoclonal |
| Clone Number | 5C4.B8 (Nivolumab) |
| Tested applications | Blocking, FC, IHC, SBR |
| Reactivity | Human, Monkey |
| Isotype | IgG |
| Immunogen | mAb PD1.5 was prepared by immunizing IgH and IgK knock-out transgenic mice possessing a human immunoglobulin (heavy chain) minilocus with recombinant human PD-1-Fc protein consisting of the extracellular domain of PD-1 (amino acids 1–167) and the Fc portion of human IgG1, and Chinese hamster ovary (CHO) cells expressing human PD-1. Nivolumab was generated by grafting the variable regions of PD1.5 onto human kappa and IgG4 constant regions containing an S228P mutation (prevents Fab arm exchange with endogenous IgG4 antibodies). |
| Concentration | 1 mg/ml |
| Conjugation | Unconjugated |
| Target | PD-1 |
| UniProt ID | Q15116 |
| Storage | Store at 4°C for up to 3 months. For longer storage, aliquot and store at -20°C. |
| Buffer/Preservatives | PBS with 0.02% Proclin 300. |
| Alternative names | CD279; programmed death-1; programmed death 1 ; PD Read more... |
| Note | For research use only |
| Application notes | Nivolumab has been shown to bind to PD-1-expressing CHO cells (EC50 ~1.66nM). Nivolumab binds CD4+ T cells (EC50 ~0.64 nM) and stains only memory and effector, and not naiive CD4+ or CD8+ T cells from human peripheral blood by FC. The antibody is able to block the interaction between PD-1 and its ligands PDL-1 and PDL-2 (IC50 ~2.52 nM and ~2.59 nM, respectively - determined by SPR) - these IC50 values are also similar to that measured by FACS to evaluate ligand binding to PD-1 expressed on CHO cells. In an allogenic T-cell/DC MLR, Nivolumab-mediated inhibition of PD-1 results in enhancement of IFNγ release, and also enhances IL-2 secretion (97-139% over an isotype control) in response to the superantigen SEB using human peripheral blood mononuclear cells. The same is also observed in a CMV-restimulation assay. Nivolumab at very low concentrations (∼1.5 ng/mL) is able to enhance T-cell reactivity in the presence of a T-cell receptor stimulus - nivolumab has no stimulatory effect in the absence of antigen or T-cell receptor stimulus. In the therapeutically used human IgG4 (S228P) format, this antibody is unable to mediate ADCC (antibody-dependent cell-mediated cytotoxicity) or CDC (complement-dependent cytotoxicity). |
| Expiration Date | 12 months from date of receipt. |
PDCD1LG2 Antibody [orb1239861]
ELISA, IF, IHC-P, WB
Rat
Human, Mouse
Rabbit
Polyclonal
Unconjugated
0.1 mg, 0.02 mgPD-L1 Antibody / B7-H1 / CD274 [orb606675]
ELISA, FACS, IF, IHC-P, WB
Human
Mouse
Monoclonal
Unconjugated
100 μg, 20 μg
Submit a review
Filter by Rating
- 5 stars
- 4 stars
- 3 stars
- 2 stars
- 1 stars