You have no items in your shopping cart.
Tips for Optimizing your IF Staining
What is IF (Immunofluorescence) staining?
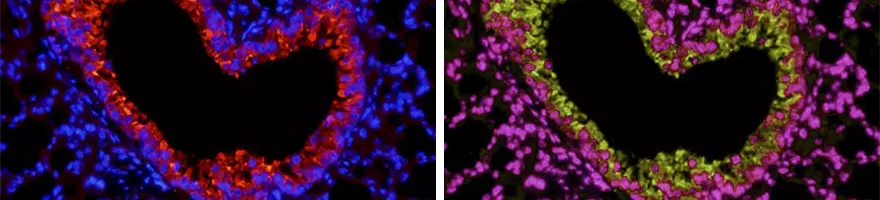
Immunofluorescence (IF) is a powerful tool used to detect and identify the subcellular distribution and movement of proteins and other molecules within cells and tissues. IF can be used as an alternative to traditional chromogenic labeling used in Immunohistochemistry (IHC) or Immunocytochemistry (ICC). By using multiple secondary antibodies, the colocalization of proteins of interest can be studied, which cannot be achieved through conventional IHC or ICC. This is an advantage when analyzing many targets at the same time, generating a high volume of accurate data. Please see our [Immunofluorescence protocol] for a step-by-step guide.
When to use IF
IF is an excellent tool to illustrate expression, localization, distribution and movement of products of interest such as molecules, proteins or glycoproteins.
IF is used as an alternative to chromogenic labelings, such as 3,3'-Diaminobenzidine (DAB).
The difference between IF & ICC & IHC
IHC and ICC differ in the type of sample used. IHC utilizes whole tissue samples which are usually frozen or paraffin-embedded. ICC refers to either isolated or cultured cells instead. Both methods can use IF as a labeling mechanism. Direct IF uses a primary antibody with an attached fluorescent dye (a conjugated antibody). Indirect IF uses a primary antibody that binds to the target and a secondary conjugated antibody that binds to the primary.
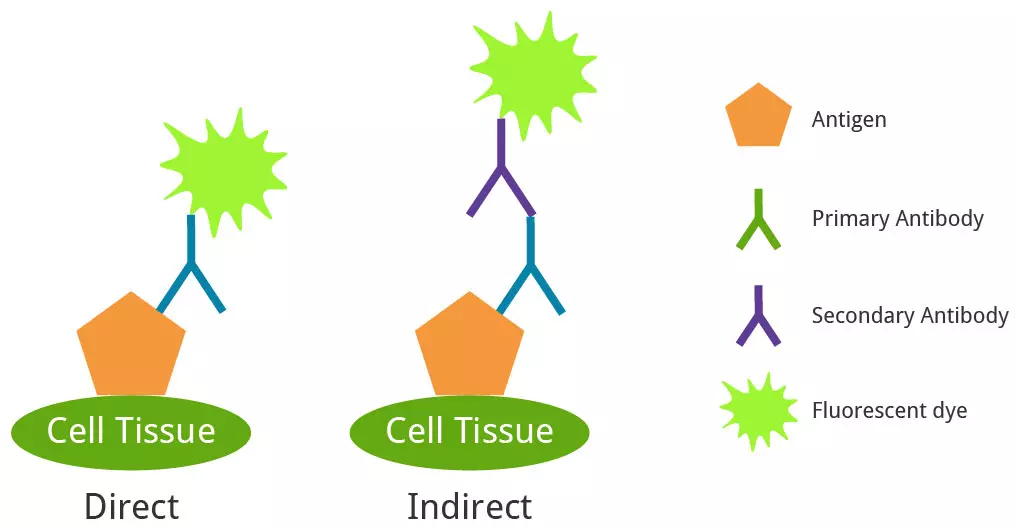
Direct and indirect IF.
Troubleshooting IF Issues
| Cause | Solution |
|---|---|
| Non-Specific Staining |
Check for spectral overlap of fluorophores used
|
| Weak Staining |
Can be caused by a variety of reasons.
|
| Background Signal |
Reagent or Sample issue.
|
Optimizing IF
These tips and tricks will save you both time and costs by optimizing each step, which will prevent wasting your valuable samples.
Include controls: Both positive and negative controls can be used to ascertain the accuracy of your results and help to work out where any issues may originate from.
Sample and reagent storage: Due to the fluorescent nature of the antibodies used and the samples produced for IF, it is essential to store both appropriately. It’s best to store both the fluorescent antibodies and the IF-treated samples in complete darkness. Use amber colored or foil covered vials for the antibodies.
Use an appropriate fixative: Aldehydes can be used for labeling of membrane-bound or cytoskeletal antigens and should be used for nuclear or mitochondrial proteins. Aldehyde fixation requires a permeabilization step for antigen detection to work correctly. Organic solvents are recommended for monoclonal antibody use. Methanol is excellent for frozen samples; Acetone is better for histological preservation.
Choose antibodies carefully: All antibodies, whether monoclonal or polyclonal, should be checked to ensure a high affinity with the antigen of interest. The secondary antibody used must be compatible with the primary, i.e. detect a mouse primary antibody using an anti-mouse secondary.
Multi-color staining: It's best to use primary antibodies raised in different species. If using indirect detection, use secondary antibodies that have been pre-adsorbed against the host species of the primary and secondary antibodies detecting other proteins, as well as the species of the experimental sample. This minimizes cross-species reactivity and non-specific binding. All secondary antibodies should also be raised in the same host species to minimize cross-reactivity. Once specificity has been established, the antigens of interest can be incubated with their antibodies simultaneously or sequentially. Sequential incubations tend to generate better results. For colocalization or counterstaining of subcellular targets, choose antibodies specific to the organelle marker.
Decide on direct or indirect staining: Both methods have advantages and disadvantages. With both methods, make sure to perform a thorough washing to minimize non-specific binding.
Choose fluorophores with no spectral overlap: IF is an excellent technique for multiplexing, with so many parameters being measured, it is essential to use fluorophores that will provide discernable signals.