You have no items in your shopping cart.
GPCR Signaling Pathways & Mechanisms
A comprehensive, researcher-focused guide to G-protein coupled receptors, downstream second messengers, and advanced strategies for recombinant membrane protein expression.
G-protein-coupled receptors (GPCRs) form the largest and most versatile family of membrane receptors in eukaryotic cells. Acting as the cellular "inbox," they process external stimuli ranging from light and odors to neurotransmitters and hormones translating them into vital intracellular responses.
1. Target Overview: The G-Protein Mechanism
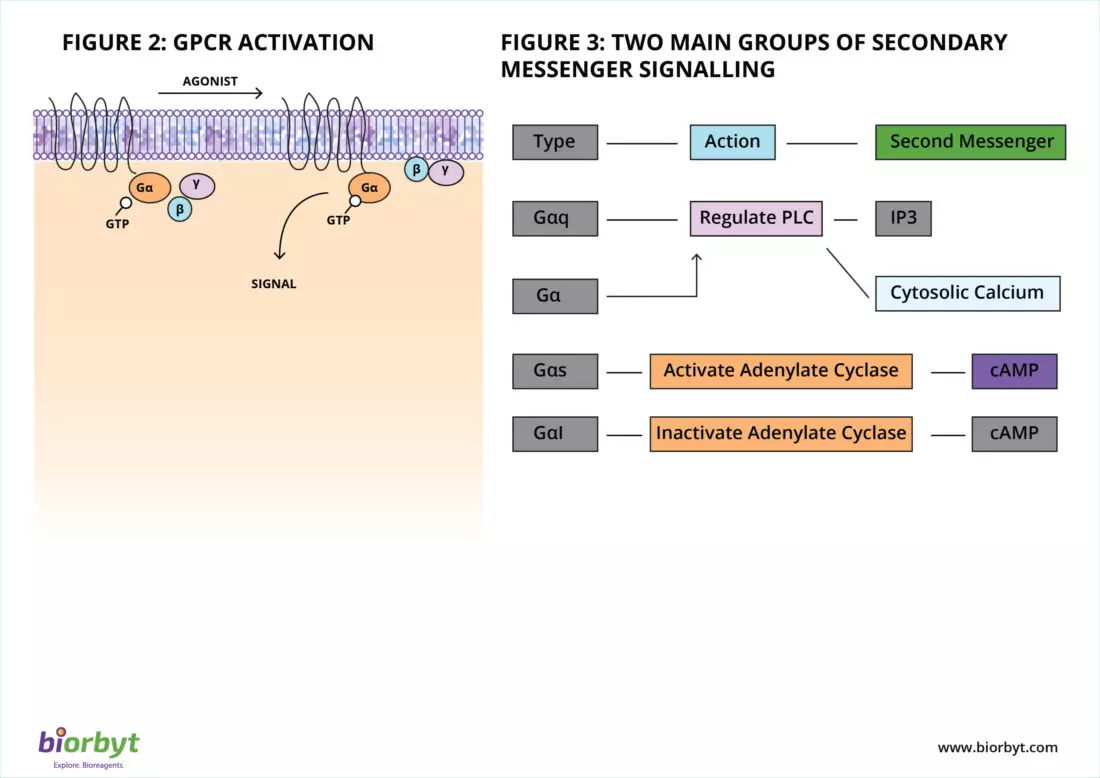
At the heart of the GPCR signal transduction pathway are heterotrimeric G proteins. These specialized, membrane-anchored proteins act as a molecular switchboard. They are composed of three distinct subunits: an alpha (α), a beta (β), and a gamma (γ) subunit . In their resting state, they are bound to the nucleotide guanosine diphosphate (GDP).
When a ligand binds to the extracellular pocket of the GPCR, the receptor undergoes a 3D structural shift. This conformational change activates the associated G-protein by prompting it to swap GDP for energy-rich GTP . The activated G-protein then splits into two distinct, functional components: the active α-subunit and the β/γ-complex. These subunits travel across the plasma membrane to interact with specific enzymes or ion channels, kicking off a powerful cellular cascade.
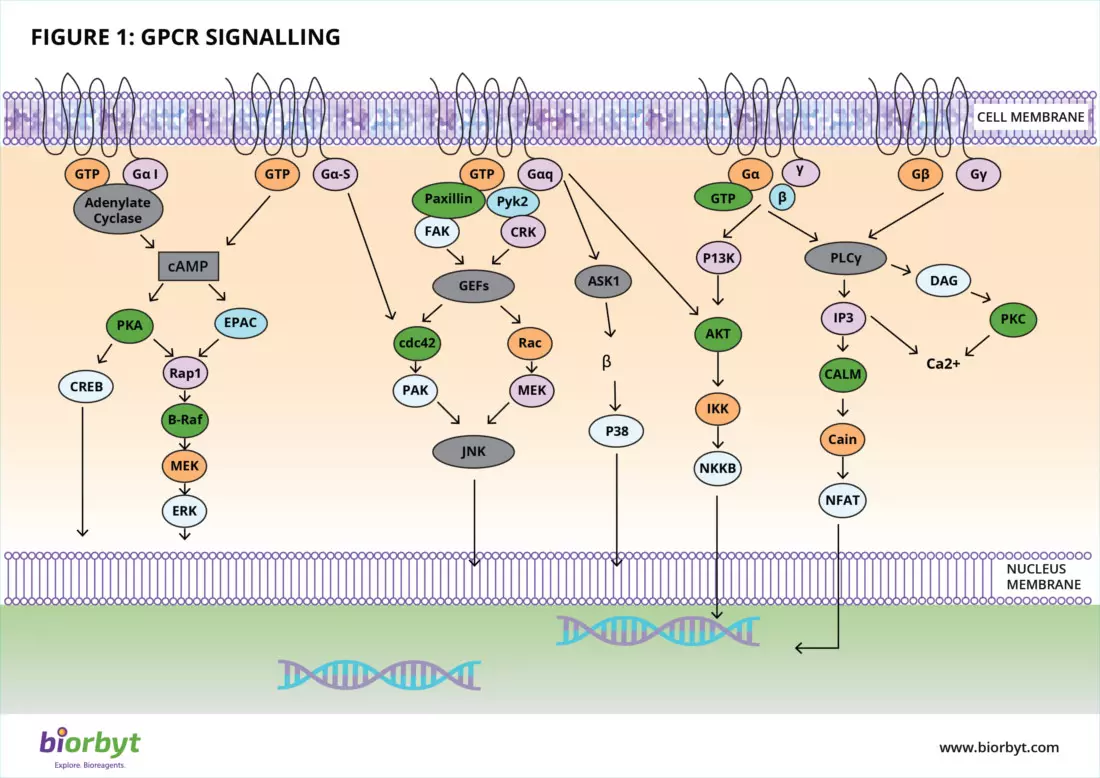
2. Generating Second Messengers
Once the G-protein subunits dissociate, they hunt for their specific effector enzymes. The cellular response that follows depends entirely on which sub-class of G-protein was activated. The two primary signaling cascades involve regulating cAMP and calcium levels:
- The Adenylate Cyclase Pathway (Gαs and Gαi): The Gαs subunit actively stimulates the adenylate cyclase enzyme to convert ATP into cyclic AMP (cAMP). This rapid surge in cAMP activates downstream targets like Protein Kinase A (PKA). Conversely, the Gαi subunit acts as the brakes, inhibiting adenylate cyclase to lower cAMP levels.
- The Phospholipase C Pathway (Gαq/11): Other subunits, primarily Gαq, activate the enzyme Phospholipase C (PLC). PLC cleaves a membrane lipid known as PIP2 into two potent secondary messengers: DAG and IP3. While DAG activates Protein Kinase C at the membrane, IP3 diffuses into the cytosol, binding to the endoplasmic reticulum to release a flood of stored calcium ions (Ca2+).
3. Physiological and Pathological Significance
Because GPCRs are capable of binding a staggering variety of ligands—including biogenic amines, peptides, lipids, and sensory cues—they govern almost every essential physiological function in the human body. Naturally, when GPCR signaling goes awry, it leads to disease.
Clinical Target Relevance: Dysregulation of GPCRs is heavily implicated in chronic pain, asthma, cardiovascular disease, central nervous system disorders, and oncology. Because of their accessible position on the cell surface and their profound impact on biology, GPCRs currently represent the target of over 30% of all FDA-approved drugs on the market today.
4. Advanced Laboratory Workflows & Assays
Studying GPCR signal transduction requires highly sensitive molecular techniques. Modern pharmacologists and life science researchers rely heavily on these functional assays:
cAMP & Calcium Flux Quantification
To validate whether a novel compound acts as a GPCR agonist or antagonist, researchers must measure the direct result of G-protein activation. Highly sensitive reporter assays, utilizing FRET or specialized ELISAs, allow labs to quantify intracellular cAMP or Ca2+ levels in real-time.
β-Arrestin Recruitment & Desensitization
To prevent cellular exhaustion, activated GPCRs are quickly phosphorylated by kinases (GRKs), which subsequently recruit β-arrestin proteins. This process blocks further G-protein signaling and triggers receptor internalization. BRET (Bioluminescence Resonance Energy Transfer) assays are universally used to measure this dynamic physical recruitment.
5. Protein QC & In-Vitro Expression Challenges
While assaying downstream second messengers is straightforward, studying the physical GPCR receptor itself in vitro is one of the greatest challenges in biochemistry. Generating stable, functional recombinant GPCRs presents several major hurdles:
Structural Challenge | Impact on Lab Research |
|---|---|
7-Transmembrane (7TM) Domains | The core of a GPCR is incredibly hydrophobic. Extracting them from the native lipid bilayer frequently causes immediate misfolding and aggregation in standard aqueous buffers. |
Conformational Flexibility | GPCRs are highly dynamic. Without precise stabilization using lipid nanodiscs or optimized detergent micelles, they rapidly lose their active 3D shape and their ligand-binding capabilities. |
Glycosylation Requirements | Bacterial expression systems (like E. coli) cannot provide the post-translational sugar modifications necessary for a GPCR's extracellular domain to fold correctly. |
6. Buying Guide & Custom Expression Solutions
Because of their inherent instability, many laboratories struggle to express functional GPCRs in-house. To ensure your binding kinetics or antibody generation workflows yield reproducible, artifact-free data, it is critical to source your proteins correctly.
Struggling to Express GPCRs?
Stop wasting months on optimization. Biorbyt’s specialized protein team can deliver highly pure, functionally active targets designed exactly for your workflow. Choose soluble Extracellular Domains (ECDs) for high-throughput antibody screening, or Full-Length Membrane Proteins stabilized in Nanodiscs for complex structural biology and SPR.
7. Companion Bioreagents
Beyond custom expression, Biorbyt offers an expansive catalog of rigorously validated tools to map and measure GPCR signaling cascades accurately.
8. Scientific Bibliography
- Shchepinova MM, Hanyaloglu AC, Frost GS, Tate EW (2020) Chemical biology of noncanonical G protein-coupled receptor signaling: Toward advanced therapeutics.. Annual Review of Biochemistry, 87, 897-919. doi:10.1016/j.cbpa.2020.04.012.
- Weis, W. I., & Kobilka, B. K. (2018). The Molecular Basis of G Protein–Coupled Receptor Activation. Annual Review of Biochemistry, 87, 897-919. doi:0.1146/annurev-biochem-060614-033910
- Samuel Liu, Preston J. et al (2024). G Protein-Coupled Receptors: A Century of Research and Discovery. Nature, 459(7245), 356-363. doi:10.1038/nature08144
- Zhang, M., Chen, T., Lu, X. et al. (2024).G protein-coupled receptors (GPCRs): advances in structures, mechanisms and drug discovery. Sig Transduct Target Ther9, 88, 3(9), 639-650. doi:10.1038/s41392-024-01803-6
