You have no items in your shopping cart.
Covid-19 ELISA Assays
Biorbyt and Daresbury Proteins are excited to announce the launch of novel Covid-19 IgG and IgM ELISA assays, providing sensitive and reliable measurements of specific antibodies as a result of Covid-19 infections and vaccination programmes.
We manufacture trimeric Spike proteins — the most valuable Covid-19 antigen — ensuring the highest sensitivity of the resulting test. They are produced in human cell culture and therefore are the exact match of the current vaccines, providing the best tool to assess the efficacy of those vaccines.
Uniquely expressed large MW proteins with extensive post-translational modifications via codon optimised sequences in proprietary expression plasmids.
Super stable trimeric Spikes as abundant capture antigens ensuring the most faithful detection of circulating specific anti-SARS-CoV-2 antibodies in recovered or vaccinated people.
Advantages
- Small volumes of blood required which can be collected at home.
- Extremely sensitive and reliable antibody-measuring method.
- Minimum consumables and instruments required to run.
- Distinguishes between serological response to various mutants/variants of concern; has a potential prognostic value.
Disadvantages of alternative antibody assays
- Most of the current commercial diagnostic kits have either recombinant Nucleocapsid or much smaller fragments of Spike as their components.
- The former cannot distinguish between SARS-CoV-2 and other coronaviruses such as flu or common cold, presenting specificity issues of the resulting tests. Moreover, it cannot be used to measure SARS-CoV-2 specific antibodies in the blood of vaccinated people.
- The Receptor Binding Domain (RBD) based tests cause sensitivity problems since they can only detect a small portion of specific antibodies.
- Prone to failure when measuring serological response to other mutant forms of Spike due to neutralisation escape effect.
Quantitative IgG & IgM ELISA
The COVID-19 ELISA IgG test is available in two formats:
- Format 1: A 96 well plate with coated Spike protein at a concentration of 2 ug/ml in each well and all required reagents to run an ELISA protocol. A thoroughly tested protocol is provided. Expiration date is 7 days from the receipt of package.
- Format 2: All reagents are provided to end user. Plate coating is carried out within the end user environment, allowing long term storage of the COVID-19 Spike protein.
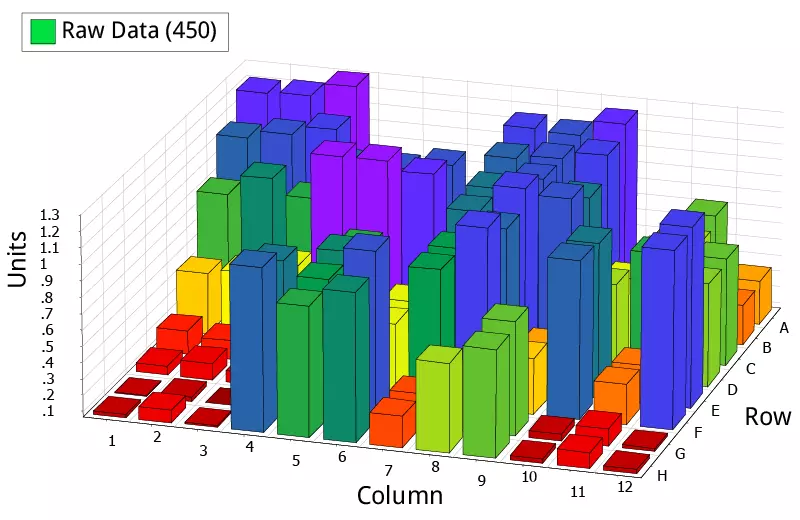
ELISA plate: raw OD data for positive and negative samples.
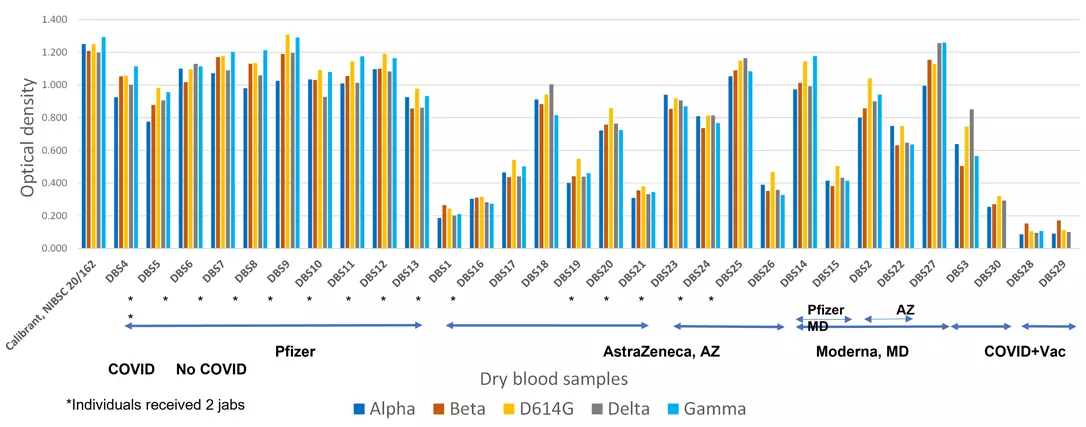
Individual immune responses to different Variants of Concern resulting from vaccinations and Covid-19 infection. Dry blood sample assay.
Isolated recombinant proteins of the various COVID-19 variants
|
|
|
|
|---|---|---|---|
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
|







