You have no items in your shopping cart.
Antibodies - Chicken Ig Why?
Immunoglobulin Y (IgY) is the dominant immunoglobulin found in avian, amphibian and reptilian species. In 1893, Klemperer1 first described the concept of passive immunity by reporting the presence and transference of tetanus toxin immunity (antibodies) from hen to chick via egg. He noted that antibodies were present in both the blood of the chicken and the yolk of the egg that it laid. However, it wasn’t until 1969 that Leslie and Clem2 coined the term IgY to describe poultry antibodies, including those found in the yolk of the eggs. Until 1969, IgY was known and reported as IgG, but the structural and antigenic differences were shown to be great enough to earn the protein the new Immunoglobulin classification IgY (Figure 1).
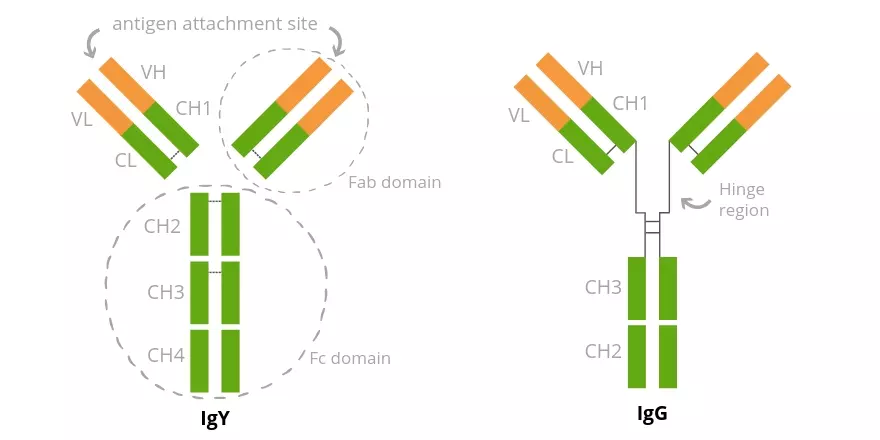
(Figure 1) Avian IgY structure, compared to mammalian IgG. Both contain two heavy and two light chains, which consist of a variable domain (VH and VL) and four constant domains (CH1, CH2, CH3 and CH4). IgG has a longer hinge region making it more flexible than avian IgY
IgG and IgY are similar on several levels:
Both have the characteristic antibody Y shape
Both contain 2 heavy and 2 light chains
Both are divalent
Both play a similar biological role, being the major immunoglobulins to provide defense against infectious agents, appearing in high concentrations after initial synthesis of the higher molecular weight antibody (IgM)
IgY also shares some similarities with mammalian IgE, including similar intrachain disulphide bonding in their extra heavy chain domain. This led to suggestions that IgY may well be an ancestral molecule to both mammalian IgG and IgE, which have since been confirmed by genetic and structural studies3 4 5.
IgG and IgY are different on several levels:
Little or no immunological cross-reactivity between IgY and mammalian IgG
IgY - higher molecular weight due to an extra heavy chain constant domain
IgY lacks a well-defined hinge region
IgY heavy chains lack Fc domains so they don’t fix complement or bind protein A or protein G
IgY can facilitate anaphylactic reactions, which can only be done by IgE in mammals
So, given these differences and similarities, why would you want to use IgYs in your research?
The very fact that the molecules are so similar in both their biological role and their structure, yet different enough not to cross-react offers several advantages to a research scientist. In most experimental applications, including Western blotting, immunohistochemistry, immunocytochemistry, ELISA and functional blocking experiments, both IgG and IgY are equivalent, meaning IgY can generally be used as a direct alternative to IgG5.
Advantages of IgY
Higher avidity - Chickens are not mammals so their reaction to a mammalian antigen is enhanced leading to high-avidity antibodies. The response is particularly to strong to mammalian antigens and especially those which are highly conserved.
Humane – What could be more humane? The chicken lays the egg, so there is no need to bleed or otherwise conduct procedures on the chicken; you just collect the eggs. Plus, Chickens are cheaper to keep than rabbits!6
High yield – one chicken can produce large quantities of IgY against low quantities of antigen. Some can produce 2.5 - 3 g of IgY per month, which is 10-20 times the amount of a rabbit6
Quick – Compared to rabbits, you can obtain high-titre antibody from eggs as early as day 25.
Lower background - IgY does not bind to IgG Fc receptors meaning you get less false positive staining. They also do not activate mammalian complement systems.
Cross-reactivity – As there is none to mammalian species, they make an excellent addition to multiple labelling or multi-plex experiments.
Convenient – IgY is packed neatly in eggs, which can be stored for long periods and the IgY purified to the desired titre/avidity months later.
IgY can still be digested by papain to produce a divalent Fab fragment.
IgY can be conjugated to enzymes or fluorophores, biotinylated and gold–labeled by standard procedures, offering you the full range of functionality you would expect from an antibody tool.
References
- Klemperer, F. Ueber natürliche Immunität und ihre Verwerthung für die Immunisirungstherapie. Arch. Für Exp. Pathol. Pharmakol. 31, 356–382 (1893).https://link.springer.com/article/10.1007/BF01832882
- Leslie, G. A. & Clem, L. W. Phylogen of immunoglobulin structure and function. 3. Immunoglobulins of the chicken. J. Exp. Med. 130, 1337–1352 (1969).https://pubmed.ncbi.nlm.nih.gov/5352783/
- Zhang, X., Calvert, R. A., Sutton, B. J. & Doré, K. A. IgY: a key isotype in antibody evolution: IgY antibody. Biol. Rev. 92, 2144–2156 (2017).https://pubmed.ncbi.nlm.nih.gov/28299878/
- Taylor, A. I., Fabiane, S. M., Sutton, B. J. & Calvert, R. A. The Crystal Structure of an Avian IgY-Fc Fragment Reveals Conservation with both Mammalian IgG and IgE . Biochemistry 48, 558–562 (2009).https://pubmed.ncbi.nlm.nih.gov/19115948/
- Lee, W., Syed Atif, A., Tan, S. C. & Leow, C. H. Insights into the chicken IgY with emphasis on the generation and applications of chicken recombinant monoclonal antibodies. J. Immunol. Methods 447, 71–85 (2017).https://pubmed.ncbi.nlm.nih.gov/28502720/
- Amro, W. A., Al-Qaisi, W. & Al-Razem, F. Production and purification of IgY antibodies from chicken egg yolk. J. Genet. Eng. Biotechnol. 16, 99–103 (2018).https://pubmed.ncbi.nlm.nih.gov/30647711/

